Response to Biologic Drugs in Patients With Rheumatoid Arthritis and Antidrug Antibodies
- PMID: 37436748
- PMCID: PMC10339150
- DOI: 10.1001/jamanetworkopen.2023.23098
Response to Biologic Drugs in Patients With Rheumatoid Arthritis and Antidrug Antibodies
Abstract
Importance: There are conflicting data on the association of antidrug antibodies with response to biologic disease-modifying antirheumatic drugs (bDMARDs) in rheumatoid arthritis (RA).
Objective: To analyze the association of antidrug antibodies with response to treatment for RA.
Design, setting, and participants: This cohort study analyzed data from the ABI-RA (Anti-Biopharmaceutical Immunization: Prediction and Analysis of Clinical Relevance to Minimize the Risk of Immunization in Rheumatoid Arthritis Patients) multicentric, open, prospective study of patients with RA from 27 recruiting centers in 4 European countries (France, Italy, the Netherlands, and the UK). Eligible patients were 18 years or older, had RA diagnosis, and were initiating a new bDMARD. Recruitment spanned from March 3, 2014, to June 21, 2016. The study was completed in June 2018, and data were analyzed in June 2022.
Exposures: Patients were treated with a new bDMARD: adalimumab, infliximab (grouped as anti-tumor necrosis factor [TNF] monoclonal antibodies [mAbs]), etanercept, tocilizumab, and rituximab according to the choice of the treating physician.
Main outcomes and measures: The primary outcome was the association of antidrug antibody positivity with EULAR (European Alliance of Associations for Rheumatology; formerly, European League Against Rheumatism) response to treatment at month 12 assessed through univariate logistic regression. The secondary end points were the EULAR response at month 6 and at visits from month 6 to months 15 to 18 using generalized estimating equation models. Detection of antidrug antibody serum levels was performed at months 1, 3, 6, 12, and 15 to 18 using electrochemiluminescence (Meso Scale Discovery) and drug concentration for anti-TNF mAbs, and etanercept in the serum was measured using enzyme-linked immunosorbent assay.
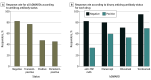
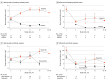
Results: Of the 254 patients recruited, 230 (mean [SD] age, 54.3 [13.7] years; 177 females [77.0%]) were analyzed. At month 12, antidrug antibody positivity was 38.2% in patients who were treated with anti-TNF mAbs, 6.1% with etanercept, 50.0% with rituximab, and 20.0% with tocilizumab. There was an inverse association between antidrug antibody positivity (odds ratio [OR], 0.19; 95% CI, 0.09-0.38; P < .001) directed against all biologic drugs and EULAR response at month 12. Analyzing all the visits starting at month 6 using generalized estimating equation models confirmed the inverse association between antidrug antibody positivity and EULAR response (OR, 0.35; 95% CI, 0.18-0.65; P < .001). A similar association was found for tocilizumab alone (OR, 0.18; 95% CI, 0.04-0.83; P = .03). In the multivariable analysis, antidrug antibodies, body mass index, and rheumatoid factor were independently inversely associated with response to treatment. There was a significantly higher drug concentration of anti-TNF mAbs in patients with antidrug antibody-negative vs antidrug antibody-positive status (mean difference, -9.6 [95% CI, -12.4 to -6.9] mg/L; P < 001). Drug concentrations of etanercept (mean difference, 0.70 [95% CI, 0.2-1.2] mg/L; P = .005) and adalimumab (mean difference, 1.8 [95% CI, 0.4-3.2] mg/L; P = .01) were lower in nonresponders vs responders. Methotrexate comedication at baseline was inversely associated with antidrug antibodies (OR, 0.50; 95% CI, 0.25-1.00; P = .05).
Conclusions and relevance: Results of this prospective cohort study suggest an association between antidrug antibodies and nonresponse to bDMARDs in patients with RA. Monitoring antidrug antibodies could be considered in the treatment of these patients, particularly nonresponders to biologic RA drugs.
Conflict of interest statement
Figures

References
-
- Gottenberg JE, Morel J, Perrodeau E, et al. ; French Society of Rheumatology and the investigators participating in AIR, ORA, and REGATE registries . Comparative effectiveness of rituximab, abatacept, and tocilizumab in adults with rheumatoid arthritis and inadequate response to TNF inhibitors: prospective cohort study. BMJ. 2019;364:l67. doi:10.1136/bmj.l67 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

