Protein-Based Model for Energy Transfer between Photosynthetic Light-Harvesting Complexes Is Constructed Using a Direct Protein-Protein Conjugation Strategy
- PMID: 37438911
- PMCID: PMC10375525
- DOI: 10.1021/jacs.3c02577
Protein-Based Model for Energy Transfer between Photosynthetic Light-Harvesting Complexes Is Constructed Using a Direct Protein-Protein Conjugation Strategy
Abstract
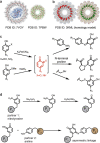
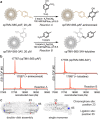
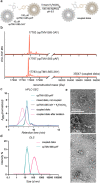
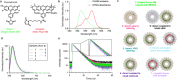
Photosynthetic organisms utilize dynamic and complex networks of pigments bound within light-harvesting complexes to transfer solar energy from antenna complexes to reaction centers. Understanding the principles underlying the efficiency of these energy transfer processes, and how they may be incorporated into artificial light-harvesting systems, is facilitated by the construction of easily tunable model systems. We describe a protein-based model to mimic directional energy transfer between light-harvesting complexes using a circular permutant of the tobacco mosaic virus coat protein (cpTMV), which self-assembles into a 34-monomer hollow disk. Two populations of cpTMV assemblies, one labeled with donor chromophores and another labeled with acceptor chromophores, were coupled using a direct protein-protein bioconjugation method. Using potassium ferricyanide as an oxidant, assemblies containing o-aminotyrosine were activated toward the addition of assemblies containing p-aminophenylalanine. Both of these noncanonical amino acids were introduced into the cpTMV monomers through amber codon suppression. This coupling strategy has the advantages of directly, irreversibly, and site-selectively coupling donor with acceptor protein assemblies and avoids cross-reactivity with native amino acids and undesired donor-donor or acceptor-acceptor combinations. The coupled donor-acceptor model was shown to transfer energy from an antenna disk containing donor chromophores to a downstream disk containing acceptor chromophores. This model ultimately provides a controllable and modifiable platform for understanding photosynthetic interassembly energy transfer and may lead to the design of more efficient functional light-harvesting materials.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
Similar articles
-
Towards building artificial light harvesting complexes: enhanced singlet-singlet energy transfer between donor and acceptor pairs bound to albumins.Photochem Photobiol Sci. 2008 Dec;7(12):1522-30. doi: 10.1039/b811091c. Epub 2008 Oct 16. Photochem Photobiol Sci. 2008. PMID: 19037505
-
Biohybrid photosynthetic antenna complexes for enhanced light-harvesting.J Am Chem Soc. 2012 Mar 14;134(10):4589-99. doi: 10.1021/ja207390y. Epub 2012 Feb 29. J Am Chem Soc. 2012. PMID: 22375881
-
Light Absorption and Energy Transfer in the Antenna Complexes of Photosynthetic Organisms.Chem Rev. 2017 Jan 25;117(2):249-293. doi: 10.1021/acs.chemrev.6b00002. Epub 2016 Jul 18. Chem Rev. 2017. PMID: 27428615 Review.
-
Energy transfer dynamics and the mechanism of biohybrid photosynthetic antenna complexes chemically linked with artificial chromophores.Phys Chem Chem Phys. 2022 Oct 19;24(40):24714-24726. doi: 10.1039/d2cp02465a. Phys Chem Chem Phys. 2022. PMID: 36128743
-
Photosynthetic Light-Harvesting (Antenna) Complexes-Structures and Functions.Molecules. 2021 Jun 3;26(11):3378. doi: 10.3390/molecules26113378. Molecules. 2021. PMID: 34204994 Free PMC article. Review.
Cited by
-
Development of artificial photosystems based on designed proteins for mechanistic insights into photosynthesis.Protein Sci. 2024 Oct;33(10):e5164. doi: 10.1002/pro.5164. Protein Sci. 2024. PMID: 39276008 Review.
-
Heteromeric guanosine (G)-quadruplex derived antenna modules with directional energy transfer.Nanoscale. 2023 Dec 7;15(47):19069-19073. doi: 10.1039/d3nr04086k. Nanoscale. 2023. PMID: 37990645 Free PMC article.
-
Virus nanotechnology for intratumoural immunotherapy.Nat Rev Bioeng. 2024 Nov;2(11):916-929. doi: 10.1038/s44222-024-00231-z. Epub 2024 Sep 23. Nat Rev Bioeng. 2024. PMID: 39698315 Free PMC article.
References
-
- MacGregor-Chatwin C.; Jackson P. J.; Şener M.; Chidgey J. W.; Hitchcock A.; Qian P.; Mayneord G. E.; Johnson M. P.; Luthey-Schulten Z.; Dickman M. J.; Scanlan D. J.; Hunter C. N. Membrane Organization of Photosystem I Complexes in the Most Abundant Phototroph on Earth. Nat. Plants 2019, 5, 879–889. 10.1038/s41477-019-0475-z. - DOI - PMC - PubMed
-
- Freer A.; Prince S.; Sauer K.; Papiz M.; Lawless A. H.; McDermott G.; Cogdell R.; Isaacs N. W. Pigment–Pigment Interactions and Energy Transfer in the Antenna Complex of the Photosynthetic Bacterium Rhodopseudomonas Acidophila. Structure 1996, 4, 449–462. 10.1016/S0969-2126(96)00050-0. - DOI - PubMed
-
- Cao P.; Bracun L.; Yamagata A.; Christianson B. M.; Negami T.; Zou B.; Terada T.; Canniffe D. P.; Shirouzu M.; Li M.; Liu L.-N. Structural Basis for the Assembly and Quinone Transport Mechanisms of the Dimeric Photosynthetic RC–LH1 Supercomplex. Nat. Commun. 2022, 13, 1977.10.1038/s41467-022-29563-3. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

