Proteomics analysis of urine and catheter-associated biofilms in spinal cord injury patients
- PMID: 37441441
- PMCID: PMC10333135
Proteomics analysis of urine and catheter-associated biofilms in spinal cord injury patients
Abstract
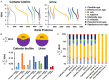
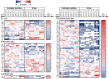
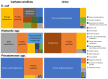
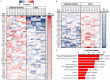
After spinal cord injury (SCI), use chronic urinary catheters for bladder management is common, making these patients especially vulnerable to catheter-associated complications. Chronic catheterization is associated with bacterial colonization and frequent catheter-associated urinary tract infections (CAUTI). One determinant of infection success and treatment resistance is production of catheter-associated biofilms, composed of microorganisms and host- and microbial-derived components. To better understand the biofilm microenvironment, we performed proteomics analysis of catheter-associated biofilms and paired urine samples from four people with SCI with chronic indwelling urinary catheters. We developed a novel method for the removal of adhered cellular components on catheters that contained both human and microbial homologous proteins. Proteins from seven microbial species were identified including: Escherichia coli, Klebsiella species (spp), Enterococcus spp, Proteus mirabilis, Pseudomonas spp, Staphylococcus spp, and Candida spp. Peptides identified from catheter biofilms were assigned to 4,820 unique proteins, with 61% of proteins assigned to the biofilm-associated microorganisms, while the remainder were human-derived. Contrastingly, in urine, only 51% were assigned to biofilm-associated microorganisms and 4,554 proteins were identified as a human-derived. Of the proteins assigned to microorganisms in the biofilm and paired urine, Enterococcus, Candida spp, and P. mirabilis had greater associations with the biofilm phase, whereas E. coli and Klebsiella had greater associations with the urine phase, thus demonstrating a significant difference between the urine and adhered microbial communities. The microbial proteins that differed significantly between the biofilm and paired urine samples mapped to pathways associated with amino acid synthesis, likely related to adaptation to high urea concentrations in the urine, and growth and protein synthesis in bacteria in the biofilm. Human proteins demonstrated enrichment for immune response in the catheter-associated biofilm. Proteomic analysis of catheter-associated biofilms and paired urine samples has the potential to provide detailed information on host and bacterial responses to chronic indwelling urinary catheters and could be useful for understanding complications of chronic indwelling catheters including CAUTIs, urinary stones, and catheter blockages.
Keywords: Catheter; biofilm; microbial proteins; proteomics; spinal cord injury; urine.
AJCEU Copyright © 2023.
Conflict of interest statement
None.
Figures
References
-
- Magill SS, Edwards JR, Bamberg W, Beldavs ZG, Dumyati G, Kainer MA, Lynfield R, Maloney M, McAllister-Hollod L, Nadle J, Ray SM, Thompson DL, Wilson LE, Fridkin SK Emerging Infections Program Healthcare-Associated Infections and Antimicrobial Use Prevalence Survey Team. Multistate point-prevalence survey of health care-associated infections. N Engl J Med. 2014;370:1198–1208. - PMC - PubMed
-
- Hooton TM, Bradley SF, Cardenas DD, Colgan R, Geerlings SE, Rice JC, Saint S, Schaeffer AJ, Tambayh PA, Tenke P, Nicolle LE Infectious Diseases Society of America. Diagnosis, prevention, and treatment of catheter-associated urinary tract infection in adults: 2009 International Clinical Practice Guidelines from the Infectious Diseases Society of America. Clin Infect Dis. 2010;50:625–663. - PubMed
-
- Stickler DJ. Bacterial biofilms in patients with indwelling urinary catheters. Nat Clin Pract Urol. 2008;5:598–608. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
