Efficacy and safety comparison of esketamine-propofol with nalbuphine-propofol for upper gastrointestinal endoscopy in children: a multi-center randomized controlled trial
- PMID: 37441574
- PMCID: PMC10333751
- DOI: 10.3389/fped.2023.1126522
Efficacy and safety comparison of esketamine-propofol with nalbuphine-propofol for upper gastrointestinal endoscopy in children: a multi-center randomized controlled trial
Abstract
Background and aims: Anesthetics such as propofol, esketamine and nalbuphine are used during the upper gastrointestinal endoscopy to achieve and maintain the desired sedation level. The aim of the study was to evaluate the effectiveness and safety of propofol-nalbuphine and propofol-esketamine in children.
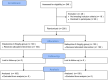
Methods: A multi-centered study was performed at three tertiary class-A hospitals. Children between 3 and 12 years old undergoing diagnostic painless upper gastrointestinal endoscopy were included and randomly divided into esketamine or nalbuphine group to estimate the primary outcome of successful endoscope insertion. The patients were given esketamine 0.5 mg/kg and propofol 2 mg/kg intravenously in esketamine group, with nalbuphine 0.2 mg/kg and propofol 2 mg/kg in the nalbuphine group. The primary outcome was success rate for the first attempt of endoscope insertion in each group. Secondary outcomes included the safety of both anesthesia regimens and gastroenterologist's satisfaction. We used the Face, Leg, Activity, Cry and Consolability (FLACC) scale to evaluate the level of pain before and during the procedure and the Pediatric Anesthesia Emergence Delirium (PAED) scale to assess the level of agitation and delirium after awakening from anesthesia.
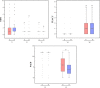
Results: Among 246 patients, 200 were randomly included in the final intention-to-treat analysis, with 100 patients in each group. The success rate for the first attempt of endoscope insertion in the esketamine group was higher than the nalbuphine group (97% vs. 66%; P < 0.01). The heart rate and mean arterial pressure after intraoperative administration in the esketamine group were higher than those in the nalbuphine group, while the delirium incidence during awakening was higher in esketamine group (all P < 0.05).
Conclusion: The success rate for the first attempt of endoscope insertion of children undergoing upper gastrointestinal endoscopy in the esketamine group was higher than the nalbuphine group, propofol-related hemodynamic changes were reduced accordingly, while the incidence of esketamine-related adverse effects could be high.
Clinical trial registration: Chinese Clinical Trial Registry: ChiCTR2000040500.
Keywords: children; esketamine; nalbuphine; painless upper gastrointestinal endoscopy; propofol.
© 2023 Zheng, Huang, Wei, Tao, Shen, Wang, He, Zhang and Sun.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
ED50 and ED95 of propofol combined with different doses of esketamine for children undergoing upper gastrointestinal endoscopy: A prospective dose-finding study using up-and-down sequential allocation method.J Clin Pharm Ther. 2022 Jul;47(7):1002-1009. doi: 10.1111/jcpt.13635. Epub 2022 Mar 7. J Clin Pharm Ther. 2022. PMID: 35255530 Clinical Trial.
-
Efficacy and safety of subanesthetic doses of esketamine combined with propofol in painless gastrointestinal endoscopy: a prospective, double-blind, randomized controlled trial.BMC Gastroenterol. 2022 Aug 20;22(1):391. doi: 10.1186/s12876-022-02467-8. BMC Gastroenterol. 2022. PMID: 35987996 Free PMC article. Clinical Trial.
-
Low-dose Esketamine combined with Propofol in microscopic pediatric strabismus surgery: a randomized controlled study.BMC Anesthesiol. 2025 May 14;25(1):241. doi: 10.1186/s12871-025-03095-x. BMC Anesthesiol. 2025. PMID: 40369437 Free PMC article. Clinical Trial.
-
Efficacy and safety of esketamine for sedation among patients undergoing gastrointestinal endoscopy: a systematic review and meta-analysis.BMC Anesthesiol. 2023 Jun 13;23(1):204. doi: 10.1186/s12871-023-02167-0. BMC Anesthesiol. 2023. PMID: 37312027 Free PMC article.
-
Efficacy and safety of low-dose esketamine for painless gastrointestinal endoscopy in adults: a systematic evaluation and meta-analysis.Front Pharmacol. 2024 Apr 5;15:1364546. doi: 10.3389/fphar.2024.1364546. eCollection 2024. Front Pharmacol. 2024. PMID: 38645560 Free PMC article.
Cited by
-
Effective doses of remimazolam and esketamine combined with remifentanil for endotracheal intubation without muscle relaxants in pediatric patients.Front Pharmacol. 2025 Mar 18;16:1558966. doi: 10.3389/fphar.2025.1558966. eCollection 2025. Front Pharmacol. 2025. PMID: 40170717 Free PMC article.
-
Intravenous esketamine as an adjuvant for sedation/analgesia outside the operating room: a systematic review and meta-analysis.Front Pharmacol. 2024 Jul 3;15:1287761. doi: 10.3389/fphar.2024.1287761. eCollection 2024. Front Pharmacol. 2024. PMID: 39021840 Free PMC article.
References
LinkOut - more resources
Full Text Sources

