Mesoporous multimetallic nanospheres with exposed highly entropic alloy sites
- PMID: 37443103
- PMCID: PMC10344865
- DOI: 10.1038/s41467-023-39157-2
Mesoporous multimetallic nanospheres with exposed highly entropic alloy sites
Erratum in
-
Publisher Correction: Mesoporous multimetallic nanospheres with exposed highly entropic alloy sites.Nat Commun. 2023 Aug 11;14(1):4862. doi: 10.1038/s41467-023-40653-8. Nat Commun. 2023. PMID: 37567887 Free PMC article. No abstract available.
Abstract
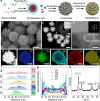
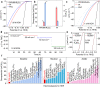
Multimetallic alloys (MMAs) with various compositions enrich the materials library with increasing diversity and have received much attention in catalysis applications. However, precisely shaping MMAs in mesoporous nanostructures and mapping the distributions of multiple elements remain big challenge due to the different reduction kinetics of various metal precursors and the complexity of crystal growth. Here we design a one-pot wet-chemical reduction approach to synthesize core-shell motif PtPdRhRuCu mesoporous nanospheres (PtPdRhRuCu MMNs) using a diblock copolymer as the soft template. The PtPdRhRuCu MMNs feature adjustable compositions and exposed porous structures rich in highly entropic alloy sites. The formation processes of the mesoporous structures and the reduction and growth kinetics of different metal precursors of PtPdRhRuCu MMNs are revealed. The PtPdRhRuCu MMNs exhibit robust electrocatalytic hydrogen evolution reaction (HER) activities and low overpotentials of 10, 13, and 28 mV at a current density of 10 mA cm-2 in alkaline (1.0 M KOH), acidic (0.5 M H2SO4), and neutral (1.0 M phosphate buffer solution (PBS)) electrolytes, respectively. The accelerated kinetics of the HER in PtPdRhRuCu MMNs are derived from multiple compositions with synergistic interactions among various metal sites and mesoporous structures with excellent mass/electron transportation characteristics.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures


Similar articles
-
Tailored Design of Mesoporous Nanospheres with High Entropic Alloy Sites for Efficient Redox Electrocatalysis.Adv Sci (Weinh). 2024 Sep;11(35):e2402518. doi: 10.1002/advs.202402518. Epub 2024 Jul 19. Adv Sci (Weinh). 2024. PMID: 39031636 Free PMC article.
-
Asymmetric Multimetallic Mesoporous Nanospheres.Nano Lett. 2019 May 8;19(5):3379-3385. doi: 10.1021/acs.nanolett.9b01223. Epub 2019 Apr 16. Nano Lett. 2019. PMID: 30974058
-
Multimetallic Hollow Mesoporous Nanospheres with Synergistically Structural and Compositional Effects for Highly Efficient Ethanol Electrooxidation.ACS Cent Sci. 2018 Oct 24;4(10):1412-1419. doi: 10.1021/acscentsci.8b00490. Epub 2018 Sep 18. ACS Cent Sci. 2018. PMID: 30410979 Free PMC article.
-
Tailored Design of Mesoporous PdCu Nanospheres with Different Compositions Using Polymeric Micelles.ACS Appl Mater Interfaces. 2019 Oct 9;11(40):36544-36552. doi: 10.1021/acsami.9b09737. Epub 2019 Sep 25. ACS Appl Mater Interfaces. 2019. PMID: 31553155
-
Preparation of Hollow Cobalt-Iron Phosphides Nanospheres by Controllable Atom Migration for Enhanced Water Oxidation and Splitting.Small. 2021 Apr;17(13):e2007858. doi: 10.1002/smll.202007858. Epub 2021 Mar 9. Small. 2021. PMID: 33690975 Review.
Cited by
-
A Regiospecific Co-Assembly Method to Functionalize Ordered Mesoporous Metal Oxides with Customizable Noble Metal Nanocrystals.ACS Cent Sci. 2024 Nov 21;10(12):2274-2284. doi: 10.1021/acscentsci.4c01592. eCollection 2024 Dec 25. ACS Cent Sci. 2024. PMID: 39735319 Free PMC article.
-
Support-Free, Connected Core-Shell Nanoparticle Catalysts Synthesized via a Low-Temperature Process for Advanced Oxygen Reduction Performance.Adv Sci (Weinh). 2025 Feb;12(7):e2408614. doi: 10.1002/advs.202408614. Epub 2024 Dec 26. Adv Sci (Weinh). 2025. PMID: 39726238 Free PMC article.
-
Nano-bio-interface: Unleashing the Potential of Noble Nanometals.Small Sci. 2024 Feb 5;4(4):2300227. doi: 10.1002/smsc.202300227. eCollection 2024 Apr. Small Sci. 2024. PMID: 40212998 Free PMC article.
-
High-entropy alloy electrocatalysts go to (sub-)nanoscale.Sci Adv. 2024 Jun 7;10(23):eadn2877. doi: 10.1126/sciadv.adn2877. Epub 2024 Jun 5. Sci Adv. 2024. PMID: 38838156 Free PMC article. Review.
-
Mesoporous amorphous non-noble metals as versatile substrates for high loading and uniform dispersion of Pt-group single atoms.Sci Adv. 2024 Jun 21;10(25):eado2442. doi: 10.1126/sciadv.ado2442. Epub 2024 Jun 21. Sci Adv. 2024. PMID: 38905333 Free PMC article.
References
-
- Ferrando R, Jellinek J, Johnston RL. Nanoalloys: from theory to applications of alloy cClusters and nanoparticles. Chem. Rev. 2008;108:845–910. - PubMed
-
- Wu Z-P, Shan S, Zang S-Q, Zhong C-J. Dynamic core–shell and alloy structures of multimetallic nanomaterials and their catalytic synergies. Acc. Chem. Res. 2020;53:2913–2924. - PubMed
-
- Yao Y, et al. High-entropy nanoparticles: synthesis-structure-property relationships and data-driven discovery. Science. 2022;376:eabn3103. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

