Ex Vivo Preservation of Ovine Periosteum Using a Perfusion Bioreactor System
- PMID: 37443758
- PMCID: PMC10340137
- DOI: 10.3390/cells12131724
Ex Vivo Preservation of Ovine Periosteum Using a Perfusion Bioreactor System
Abstract
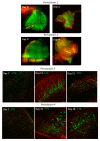
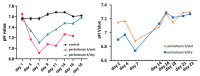
Periosteum is a highly vascularized membrane lining the surface of bones. It plays essential roles in bone repair following injury and reconstruction following invasive surgeries. To broaden the use of periosteum, including for augmenting in vitro bone engineering and/or in vivo bone repair, we have developed an ex vivo perfusion bioreactor system to maintain the cellular viability and metabolism of surgically resected periosteal flaps. Each specimen was placed in a 3D printed bioreactor connected to a peristaltic pump designed for the optimal flow rates of tissue perfusate. Nutrients and oxygen were perfused via the periosteal arteries to mimic physiological conditions. Biochemical assays and histological staining indicate component cell viability after perfusion for almost 4 weeks. Our work provides the proof-of-concept of ex vivo periosteum perfusion for long-term tissue preservation, paving the way for innovative bone engineering approaches that use autotransplanted periosteum to enhance in vivo bone repair.
Keywords: bone engineering; bone repair; perfusion; periosteum; transplantation; viability.
Conflict of interest statement
The authors of this manuscript have no conflict of interest to disclose.
Figures




References
-
- Accorona R., Gazzini L., Grigolato R., Fazio E., Nitro L., Abousiam M., Giorgetti G., Pignataro L., Capaccio P., Calabrese L. Free Periosteal Flaps with Scaffold: An Overlooked Armamentarium for Maxillary and Mandibular Reconstruction. Cancers. 2021;13:4373. doi: 10.3390/cancers13174373. - DOI - PMC - PubMed
-
- Petrides G.A., Dunn M., Charters E., Venchiarutti R., Cheng K., Froggatt C., Mukherjee P., Wallace C., Howes D., Leinkram D., et al. Health-related quality of life in maxillectomy patients undergoing dentoalveolar rehabilitation. Oral Oncol. 2022;126:105757. doi: 10.1016/j.oraloncology.2022.105757. - DOI - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

