Allyl Isothiocianate Induces Ca2+ Signals and Nitric Oxide Release by Inducing Reactive Oxygen Species Production in the Human Cerebrovascular Endothelial Cell Line hCMEC/D3
- PMID: 37443764
- PMCID: PMC10340171
- DOI: 10.3390/cells12131732
Allyl Isothiocianate Induces Ca2+ Signals and Nitric Oxide Release by Inducing Reactive Oxygen Species Production in the Human Cerebrovascular Endothelial Cell Line hCMEC/D3
Abstract
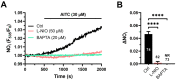
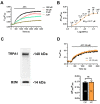
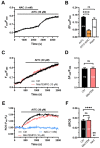
Nitric oxide (NO) represents a crucial mediator to regulate cerebral blood flow (CBF) in the human brain both under basal conditions and in response to somatosensory stimulation. An increase in intracellular Ca2+ concentrations ([Ca2+]i) stimulates the endothelial NO synthase to produce NO in human cerebrovascular endothelial cells. Therefore, targeting the endothelial ion channel machinery could represent a promising strategy to rescue endothelial NO signalling in traumatic brain injury and neurodegenerative disorders. Allyl isothiocyanate (AITC), a major active constituent of cruciferous vegetables, was found to increase CBF in non-human preclinical models, but it is still unknown whether it stimulates NO release in human brain capillary endothelial cells. In the present investigation, we showed that AITC evoked a Ca2+-dependent NO release in the human cerebrovascular endothelial cell line, hCMEC/D3. The Ca2+ response to AITC was shaped by both intra- and extracellular Ca2+ sources, although it was insensitive to the pharmacological blockade of transient receptor potential ankyrin 1, which is regarded to be among the main molecular targets of AITC. In accord, AITC failed to induce transmembrane currents or to elicit membrane hyperpolarization, although NS309, a selective opener of the small- and intermediate-conductance Ca2+-activated K+ channels, induced a significant membrane hyperpolarization. The AITC-evoked Ca2+ signal was triggered by the production of cytosolic, but not mitochondrial, reactive oxygen species (ROS), and was supported by store-operated Ca2+ entry (SOCE). Conversely, the Ca2+ response to AITC did not require Ca2+ mobilization from the endoplasmic reticulum, lysosomes or mitochondria. However, pharmacological manipulation revealed that AITC-dependent ROS generation inhibited plasma membrane Ca2+-ATPase (PMCA) activity, thereby attenuating Ca2+ removal across the plasma membrane and resulting in a sustained increase in [Ca2+]i. In accord, the AITC-evoked NO release was driven by ROS generation and required ROS-dependent inhibition of PMCA activity. These data suggest that AITC could be exploited to restore NO signalling and restore CBF in brain disorders that feature neurovascular dysfunction.
Keywords: Ca2+ signalling; allyl isothiocianate; hCMEC/D3; nitric oxide; plasma membrane Ca2+-ATPase; reactive oxygen species; store-operated Ca2+ entry.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
- National Recovery and Resilience Plan (NRRP), project MNESYS (PE0000006) - A Multiscale in-tegrated approach to the study of the nervous system in health and disease (DN. 1553 11.10.2022)./Ministry of University and Research (MUR)
- Program under Grant Agreement N. 828984, LION-HEARTED/EU Horizon 2020 FETOPEN-2018-2020
- Dipartimenti di Eccellenza Program (2018-2022) - Dept. of Biology and Biotechnology "L. Spallanzani"/Italian Ministry of Education, University and Research (MIUR)
- Fondo Ricerca Giovani/University of Pavia
- Identification Number: CVU121216/Consejo Nacional de Ciencia y Tecnología
LinkOut - more resources
Full Text Sources
Miscellaneous

