LRRK2 Knockout Confers Resistance in HEK-293 Cells to Rotenone-Induced Oxidative Stress, Mitochondrial Damage, and Apoptosis
- PMID: 37445652
- PMCID: PMC10341561
- DOI: 10.3390/ijms241310474
LRRK2 Knockout Confers Resistance in HEK-293 Cells to Rotenone-Induced Oxidative Stress, Mitochondrial Damage, and Apoptosis
Abstract
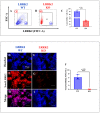
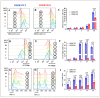
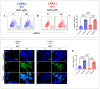
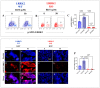
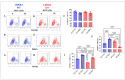
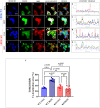
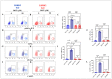
Leucine-rich repeat kinase 2 (LRRK2) has been linked to dopaminergic neuronal vulnerability to oxidative stress (OS), mitochondrial impairment, and increased cell death in idiopathic and familial Parkinson's disease (PD). However, how exactly this kinase participates in the OS-mitochondria-apoptosis connection is still unknown. We used clustered regularly interspaced short palindromic repeats (CRISPR)/Cas9 LRRK2 knockout (KO) in the human embryonic kidney cell line 293 (HEK-293) to evaluate the cellular response to the mitochondrial inhibitor complex I rotenone (ROT), a well-known OS and cell death inducer. We report successful knockout of the LRRK2 gene in HEK-293 cells using CRISPR editing (ICE, approximately 60%) and flow cytometry (81%) analyses. We found that HEK-293 LRRK2 WT cells exposed to rotenone (ROT, 50 μM) resulted in a significant increase in intracellular reactive oxygen species (ROS, +7400%); oxidized DJ-1-Cys106-SO3 (+52%); phosphorylation of LRRK2 (+70%) and c-JUN (+171%); enhanced expression of tumor protein (TP53, +2000%), p53 upregulated modulator of apoptosis (PUMA, +1950%), and Parkin (PRKN, +22%); activation of caspase 3 (CASP3, +8000%), DNA fragmentation (+35%) and decreased mitochondrial membrane potential (ΔΨm, -58%) and PTEN induced putative kinase 1 (PINK1, -49%) when compared to untreated cells. The translocation of the cytoplasmic fission protein dynamin-related Protein 1 (DRP1) to mitochondria was also observed by colocalization with translocase of the outer membrane 20 (TOM20). Outstandingly, HEK-293 LRRK2 KO cells treated with ROT showed unaltered OS and apoptosis markers. We conclude that loss of LRRK2 causes HEK-293 to be resistant to ROT-induced OS, mitochondrial damage, and apoptosis in vitro. Our data support the hypothesis that LRRK2 acts as a proapoptotic kinase by regulating mitochondrial proteins (e.g., PRKN, PINK1, DRP1, and PUMA), transcription factors (e.g., c-JUN and TP53), and CASP3 in cells under stress conditions. Taken together, these observations suggest that LRRK2 is an important kinase in the pathogenesis of PD.
Keywords: Leucine-rich repeat kinase 2; apoptosis; clustered regularly interspaced short palindromic repeats; gene edition; human embryonic kidney cell line 293; knockout; oxidative stress; rotenone.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Cornejo-Olivas M., Torres L., Velit-Salazar M.R., Inca-Martinez M., Mazzetti P., Cosentino C., Micheli F., Perandones C., Dieguez E., Raggio V., et al. Variable frequency of LRRK2 variants in the Latin American research consortium on the genetics of Parkinson’s disease (LARGE-PD), a case of ancestry. NPJ Park. Dis. 2017;3:19. doi: 10.1038/s41531-017-0020-6. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

