The Role of mTORC1 Pathway and Autophagy in Resistance to Platinum-Based Chemotherapeutics
- PMID: 37445831
- PMCID: PMC10341996
- DOI: 10.3390/ijms241310651
The Role of mTORC1 Pathway and Autophagy in Resistance to Platinum-Based Chemotherapeutics
Abstract
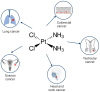
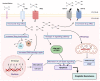
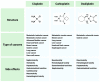
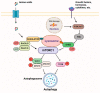
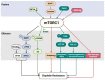
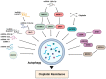
Cisplatin (cis-diamminedichloroplatinum I) is a platinum-based drug, the mainstay of anticancer treatment for numerous solid tumors. Since its approval by the FDA in 1978, the drug has continued to be used for the treatment of half of epithelial cancers. However, resistance to cisplatin represents a major obstacle during anticancer therapy. Here, we review recent findings on how the mTORC1 pathway and autophagy can influence cisplatin sensitivity and resistance and how these data can be applicable for the development of new therapeutic strategies.
Keywords: anticancer drug resistance; autophagy; cisplatin; mTORC1 pathway.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Phosphatidic acid as a cofactor of mTORC1 in platinum-based chemoresistance: Mechanisms and therapeutic potential.Eur J Pharmacol. 2025 Feb 5;988:177220. doi: 10.1016/j.ejphar.2024.177220. Epub 2024 Dec 21. Eur J Pharmacol. 2025. PMID: 39716566 Review.
-
Induction of autophagy counteracts the anticancer effect of cisplatin in human esophageal cancer cells with acquired drug resistance.Cancer Lett. 2014 Dec 1;355(1):34-45. doi: 10.1016/j.canlet.2014.09.020. Epub 2014 Sep 16. Cancer Lett. 2014. PMID: 25236911
-
Corrosion-Activated Chemotherapeutic Function of Nanoparticulate Platinum as a Cisplatin Resistance-Overcoming Prodrug with Limited Autophagy Induction.Small. 2016 Nov;12(44):6124-6133. doi: 10.1002/smll.201602374. Epub 2016 Sep 22. Small. 2016. PMID: 27717137
-
Cucurbitacin B induces autophagy and apoptosis by suppressing CIP2A/PP2A/mTORC1 signaling axis in human cisplatin resistant gastric cancer cells.Oncol Rep. 2017 Jul;38(1):271-278. doi: 10.3892/or.2017.5648. Epub 2017 May 18. Oncol Rep. 2017. PMID: 28534965
-
New-generation platinum drugs in the treatment of cisplatin-resistant cancers.Expert Opin Investig Drugs. 2005 Aug;14(8):1033-46. doi: 10.1517/13543784.14.8.1033. Expert Opin Investig Drugs. 2005. PMID: 16050795 Review.
Cited by
-
Apigenin suppresses the low oxaliplatin-induced epithelial-mesenchymal transition in oral squamous cell carcinoma cells via LINC00857.Transl Cancer Res. 2024 May 31;13(5):2164-2174. doi: 10.21037/tcr-23-2335. Epub 2024 May 29. Transl Cancer Res. 2024. PMID: 38881938 Free PMC article.
-
Introduction of AGPAT3 gene as a regulator of cisplatin resistance in A2780 ovarian endometrioid carcinoma cell line.PLoS One. 2025 Mar 10;20(3):e0318740. doi: 10.1371/journal.pone.0318740. eCollection 2025. PLoS One. 2025. PMID: 40063560 Free PMC article.
-
The m-TORC1 inhibitor Sirolimus increases the effectiveness of Photodynamic therapy in the treatment of cutaneous Squamous Cell Carcinoma, impairing NRF2 antioxidant signaling.Int J Biol Sci. 2024 Aug 6;20(11):4238-4257. doi: 10.7150/ijbs.94883. eCollection 2024. Int J Biol Sci. 2024. PMID: 39247827 Free PMC article.
-
IGF2BP3-dependent N6-methyladenosine modification of USP49 promotes carboplatin resistance in retinoblastoma by enhancing autophagy via regulating the stabilization of SIRT1.Kaohsiung J Med Sci. 2024 Dec;40(12):1043-1056. doi: 10.1002/kjm2.12902. Epub 2024 Nov 4. Kaohsiung J Med Sci. 2024. PMID: 39497328 Free PMC article.
-
Novel players in the development of chemoresistance in ovarian cancer: ovarian cancer stem cells, non-coding RNA and nuclear receptors.Cancer Drug Resist. 2024 Feb 28;7:6. doi: 10.20517/cdr.2023.152. eCollection 2024. Cancer Drug Resist. 2024. PMID: 38434767 Free PMC article. Review.
References
-
- Planells-Cases R., Lutter D., Guyader C., Gerhards N.M., Ullrich F., Elger D.A., Kucukosmanoglu A., Xu G., Voss F.K., Reincke S.M., et al. Subunit Composition of VRAC Channels Determines Substrate Specificity and Cellular Resistance to P T-based Anti-cancer Drugs. EMBO J. 2015;34:2993–3008. doi: 10.15252/embj.201592409. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

