Stealth Liposomes Encapsulating a Potent ACAT1/SOAT1 Inhibitor F12511: Pharmacokinetic, Biodistribution, and Toxicity Studies in Wild-Type Mice and Efficacy Studies in Triple Transgenic Alzheimer's Disease Mice
- PMID: 37446191
- PMCID: PMC10341764
- DOI: 10.3390/ijms241311013
Stealth Liposomes Encapsulating a Potent ACAT1/SOAT1 Inhibitor F12511: Pharmacokinetic, Biodistribution, and Toxicity Studies in Wild-Type Mice and Efficacy Studies in Triple Transgenic Alzheimer's Disease Mice
Abstract
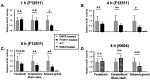
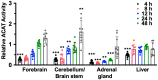
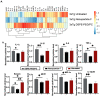
Cholesterol is essential for cellular function and is stored as cholesteryl esters (CEs). CEs biosynthesis is catalyzed by the enzymes acyl-CoA:cholesterol acyltransferase 1 and 2 (ACAT1 and ACAT2), with ACAT1 being the primary isoenzyme in most cells in humans. In Alzheimer's Disease, CEs accumulate in vulnerable brain regions. Therefore, ACATs may be promising targets for treating AD. F12511 is a high-affinity ACAT1 inhibitor that has passed phase 1 safety tests for antiatherosclerosis. Previously, we developed a nanoparticle system to encapsulate a large concentration of F12511 into a stealth liposome (DSPE-PEG2000 with phosphatidylcholine). Here, we injected the nanoparticle encapsulated F12511 (nanoparticle F) intravenously (IV) in wild-type mice and performed an HPLC/MS/MS analysis and ACAT enzyme activity measurement. The results demonstrated that F12511 was present within the mouse brain after a single IV but did not overaccumulate in the brain or other tissues after repeated IVs. A histological examination showed that F12511 did not cause overt neurological or systemic toxicity. We then showed that a 2-week IV delivery of nanoparticle F to aging 3xTg AD mice ameliorated amyloidopathy, reduced hyperphosphorylated tau and nonphosphorylated tau, and reduced neuroinflammation. This work lays the foundation for nanoparticle F to be used as a possible therapy for AD and other neurodegenerative diseases.
Keywords: ACAT1/SOAT1; Alzheimer’s disease; DSPE-PEG; F12511; amyloid; cholesterol; cholesteryl ester; nanoparticles; phosphatidylcholine; tau.
Conflict of interest statement
The authors declare no conflict of interest.
Figures






References
-
- Corder E.H., Saunders A.M., Strittmatter W.J., Schmechel D.E., Gaskell P.C., Small G.W., Roses A.D., Haines J.L., Pericak-Vance M.A. Gene dose of apolipoprotein E type 4 allele and the risk of Alzheimer’s disease in late onset families. Science. 1993;261:921–923. doi: 10.1126/science.8346443. - DOI - PubMed
-
- Beecham G.W., Hamilton K., Naj A.C., Martin E.R., Huentelman M., Myers A.J., Corneveaux J.J., Hardy J., Vonsattel J.P., Younkin S.G., et al. Genome-wide association meta-analysis of neuropathologic features of Alzheimer’s disease and related dementias. PLoS Genet. 2014;10:e1004606. doi: 10.1371/journal.pgen.1004606. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

