Quantification of Empty, Partially Filled and Full Adeno-Associated Virus Vectors Using Mass Photometry
- PMID: 37446211
- PMCID: PMC10341871
- DOI: 10.3390/ijms241311033
Quantification of Empty, Partially Filled and Full Adeno-Associated Virus Vectors Using Mass Photometry
Abstract
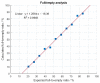
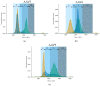
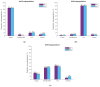
Adeno-associated viruses (AAV) are one of the most commonly used vehicles in gene therapies for the treatment of rare diseases. During the AAV manufacturing process, particles with little or no genetic material are co-produced alongside the desired AAV capsid containing the transgene of interest. Because of the potential adverse health effects of these byproducts, they are considered impurities and need to be monitored carefully. To date, analytical ultracentrifugation (AUC), transmission electron microscopy (TEM) and charge-detection mass spectrometry (CDMS) are used to quantify these subspecies. However, they are associated with long turnaround times, low sample throughput and complex data analysis. Mass photometry (MP) is a fast and label-free orthogonal technique which is applicable to multiple serotypes without the adaption of method parameters. Furthermore, it can be operated with capsid titers as low as 8 × 1010 cp mL-1 with a CV < 5% using just 10 µL total sample volume. Here we demonstrate that mass photometry can be used as an orthogonal method to AUC to accurately quantify the proportions of empty, partially filled, full and overfull particles in AAV samples, especially in cases where ion-exchange chromatography yields no separation of the populations. In addition, it can be used to confirm the molar mass of the packaged genomic material in filled AAV particles.
Keywords: adeno-associated virus vectors; analytical ultracentrifugation; genomic cargo; partially filled particles; single-molecule mass photometry.
Conflict of interest statement
Authors Christina Wagner, Felix F. Fuchsberger, Bernd Innthaler and Martin Lemmerer were employed by the company Takeda. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The authors declare that this study received funding from Baxalta Innovations GmbH (part of Takeda). The funder had the following involvement with the study: employer of Christina Wagner, Felix F. Fuchsberger, Bernd Innthaler and Martin Lemmerer.
Figures




References
-
- Wileys J. Gene Therapy Clinical Trials Worldwide. [(accessed on 12 April 2023)]. Available online: https://a873679.fmphost.com/fmi/webd/GTCT.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

