Composite Fibrin/Carbon Microfiber Implants for Bridging Spinal Cord Injury: A Translational Approach in Pigs
- PMID: 37446280
- PMCID: PMC10342401
- DOI: 10.3390/ijms241311102
Composite Fibrin/Carbon Microfiber Implants for Bridging Spinal Cord Injury: A Translational Approach in Pigs
Abstract
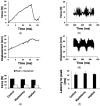
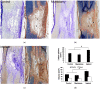
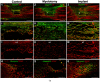
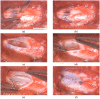
Biomaterials may enhance neural repair after spinal cord injury (SCI) and testing their functionality in large animals is essential to achieve successful clinical translation. This work developed a porcine contusion/compression SCI model to investigate the consequences of myelotomy and implantation of fibrin gel containing biofunctionalized carbon microfibers (MFs). Fourteen pigs were distributed in SCI, SCI/myelotomy, and SCI/myelotomy/implant groups. An automated device was used for SCI. A dorsal myelotomy was performed on the lesion site at 1 day post-injury for removing cloths and devitalized tissue. Bundles of MFs coated with a conducting polymer and cell adhesion molecules were embedded in fibrin gel and used to bridge the spinal cord cavity. Reproducible lesions of about 1 cm in length were obtained. Myelotomy and lesion debridement caused no further neural damage compared to SCI alone but had little positive effect on neural regrowth. The MFs/fibrin gel implant facilitated axonal sprouting, elongation, and alignment within the lesion. However, the implant also increased lesion volume and was ineffective in preventing fibrosis, thus precluding functional neural regeneration. Our results indicate that myelotomy and lesion debridement can be advantageously used for implanting MF-based scaffolds. However, the implants need refinement and pharmaceuticals will be necessary to limit scarring.
Keywords: biomaterial; compression; conducting polymer; contusion; microfiber; myelotomy; pig; porcine; regeneration; spinal cord injury.
Conflict of interest statement
J.E.C.-C. and A.A.-S. have shares in Spinal Cord Technologies S.L., a spin-off company of the Hospital Nacional de Parapléjicos in Toledo, Spain. J.E. Collazos-Castro is inventor in patent ESP201231969 regarding conducting-polymer-coated carbon MFs, and together with A. Alves-Sampaio, he is the co-inventor in the patent application P202230626 on fibrin/MFs scaffolds. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures





Similar articles
-
Biofunctionalized PEDOT-coated microfibers for the treatment of spinal cord injury.Biomaterials. 2016 May;89:98-113. doi: 10.1016/j.biomaterials.2016.02.037. Epub 2016 Feb 26. Biomaterials. 2016. PMID: 26963900
-
Intravital Assessment of Cells Responses to Conducting Polymer-Coated Carbon Microfibres for Bridging Spinal Cord Injury.Cells. 2021 Jan 5;10(1):73. doi: 10.3390/cells10010073. Cells. 2021. PMID: 33466339 Free PMC article.
-
Graphene Oxide Microfibers Promote Regenerative Responses after Chronic Implantation in the Cervical Injured Spinal Cord.ACS Biomater Sci Eng. 2020 Apr 13;6(4):2401-2414. doi: 10.1021/acsbiomaterials.0c00345. Epub 2020 Mar 24. ACS Biomater Sci Eng. 2020. PMID: 33455347
-
Biomaterial scaffolds used for the regeneration of spinal cord injury (SCI).Histol Histopathol. 2014 Nov;29(11):1395-408. doi: 10.14670/HH-29.1395. Epub 2014 May 16. Histol Histopathol. 2014. PMID: 24831814 Review.
-
Biomaterial-supported MSC transplantation enhances cell-cell communication for spinal cord injury.Stem Cell Res Ther. 2021 Jan 7;12(1):36. doi: 10.1186/s13287-020-02090-y. Stem Cell Res Ther. 2021. PMID: 33413653 Free PMC article. Review.
Cited by
-
Stability of Conducting Polymer-Coated Carbon Microfibers for Long-Term Electrical Stimulation of Injured Neural Tissue.Polymers (Basel). 2024 Jul 22;16(14):2093. doi: 10.3390/polym16142093. Polymers (Basel). 2024. PMID: 39065410 Free PMC article.
-
Functional biomaterials for modulating the dysfunctional pathological microenvironment of spinal cord injury.Bioact Mater. 2024 May 30;39:521-543. doi: 10.1016/j.bioactmat.2024.04.015. eCollection 2024 Sep. Bioact Mater. 2024. PMID: 38883317 Free PMC article. Review.
-
Electrophoretic Deposition of Calcium Phosphates on Carbon-Carbon Composite Implants: Morphology, Phase/Chemical Composition and Biological Reactions.Int J Mol Sci. 2024 Mar 16;25(6):3375. doi: 10.3390/ijms25063375. Int J Mol Sci. 2024. PMID: 38542348 Free PMC article.
References
-
- Collazos-Castro J.E. Handbook of Innovations in Central Nervous System Regenerative Medicine. Elsevier; Amsterdam, The Netherlands: 2020. Biomaterial-based systems as biomimetic agents in the repair of the central nervous system; pp. 259–289.
-
- Guest J.D., Moore S.W., Aimetti A.A., Kutikov A.B., Santamaria A.J., Hofstetter C.P., Ropper A.E., Theodore N., Ulich T.R., Layer R.T. Internal decompression of the acutely contused spinal cord: Differential effects of irrigation only versus biodegradable scaffold implantation. Biomaterials. 2018;185:284–300. doi: 10.1016/j.biomaterials.2018.09.025. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

