Recent Progress in Multifunctional Graphene-Based Nanocomposites for Photocatalysis and Electrocatalysis Application
- PMID: 37446544
- PMCID: PMC10343872
- DOI: 10.3390/nano13132028
Recent Progress in Multifunctional Graphene-Based Nanocomposites for Photocatalysis and Electrocatalysis Application
Abstract
The global energy shortage and environmental degradation are two major issues of concern in today's society. The production of renewable energy and the treatment of pollutants are currently the mainstream research directions in the field of photocatalysis. In addition, over the last decade or so, graphene (GR) has been widely used in photocatalysis due to its unique physical and chemical properties, such as its large light-absorption range, high adsorption capacity, large specific surface area, and excellent electronic conductivity. Here, we first introduce the unique properties of graphene, such as its high specific surface area, chemical stability, etc. Then, the basic principles of photocatalytic hydrolysis, pollutant degradation, and the photocatalytic reduction of CO2 are summarized. We then give an overview of the optimization strategies for graphene-based photocatalysis and the latest advances in its application. Finally, we present challenges and perspectives for graphene-based applications in this field in light of recent developments.
Keywords: CO2 fixation; electrocatalysis; graphene composites; photocatalysis; pollutant degradation.
Conflict of interest statement
The authors declared that they have no conflicts of interest to this work.
Figures




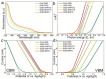

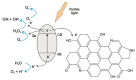
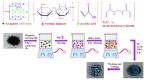

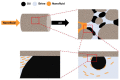






References
-
- Zhang S., Gu P., Ma R., Luo C., Wen T., Zhao G., Cheng W., Wang X. Recent developments in fabrication and structure regulation of visible-light-driven g-C3N4-based photocatalysts towards water purification: A critical review. Catal. Today. 2019;335:65–77. doi: 10.1016/j.cattod.2018.09.013. - DOI
-
- Freund H.J., Roberts M.W. Surface chemistry of carbon dioxide. Surf. Sci. Rep. 1996;25:225–273. doi: 10.1016/S0167-5729(96)00007-6. - DOI
-
- Feng J., Huang H., Yan S., Luo W., Yu T., Li Z., Zou Z. Non-oxide semiconductors for artificial photosynthesis: Progress on photoelectrochemical water splitting and carbon dioxide reduction. Nano Today. 2020;30:100830. doi: 10.1016/j.nantod.2019.100830. - DOI
-
- Ahmad A., Ali M., Al-Sehemi A.G., Al-Ghamdi A.A., Park J.-W., Algarni H., Anwer H. Carbon-integrated semiconductor photocatalysts for removal of volatile organic compounds in indoor environments. Chem. Eng. J. 2023;452:139436. doi: 10.1016/j.cej.2022.139436. - DOI
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

