Effect of Steam on Carbonation of CaO in Ca-Looping
- PMID: 37446572
- PMCID: PMC10343333
- DOI: 10.3390/molecules28134910
Effect of Steam on Carbonation of CaO in Ca-Looping
Abstract
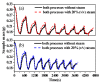
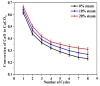
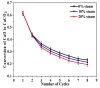
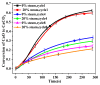
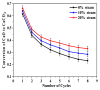
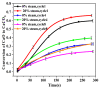
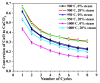
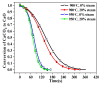
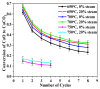
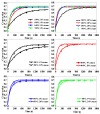
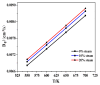
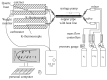
Ca-looping is an effective way to capture CO2 from coal-fired power plants. However, there are still issues that require further study. One of these issues is the effect of steam on the Ca-looping process. In this paper, a self-madethermogravimetric analyzer that can achieve rapid heating and cooling is used to measure the change of sample weight under constant temperature conditions. The parameters of the Ca-looping are studied in detail, including the addition of water vapor alone in the calcination or carbonation stage and the calcination/carbonation reaction temperatures for both calcination and carbonation stages with water vapor. Steam has a positive overall effect on CO2 capture in the Ca-looping process. When steam is present in both calcination and carbonation processes, it increases the decomposition rate of CaCO3 and enhances the subsequent carbonation conversion of CaO. However, when steam was present only in the calcination process, there was lower CaO carbonation conversion in the following carbonation process. In contrast, when steam was present in the carbonation stage, CO2 capture was improved. Sample characterizations after the reaction showed that although water vapor had a negative effect on the pore structure, adding water vapor increased the diffusion coefficient of CO2 and the carbonation conversion rate of CaO.
Keywords: Ca-looping; carbonation conversion; catalysis; steam.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Stocker T.F., Quin D. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press; Cambridge, UK: 2013. IPCC 2013: Climate Change 2013—The Physical Science Basis.
-
- Norbarzad M.J., Tahmasebpoor M., Heidari M., Pevida C. Theoretical and experimental study on the fluidity performance of hard-to-fluidize carbon nanotubes-based CO2 capture sorbents. Front. Chem. Sci. Eng. 2022;16:1460–1475. doi: 10.1007/s11705-022-2159-x. - DOI
-
- Troya J.A., Jiménez P.S.E., Perejón A., Moreno V., Valverde J.M., Maqueda P.A.P. Kinetics and cyclability of limestone (CaCO3) in presence of steam during calcination in the CaL scheme for thermochemical energy storage. Chem. Eng. J. 2021;417:129194. doi: 10.1016/j.cej.2021.129194. - DOI
-
- Han R., Wang Y., Xing S., Pang C., Hao Y., Song C., Liu Q. Progress in reducing calcination reaction temperature of Calcium-Looping CO2 capture technology: A critical review. Chem. Eng. J. 2022;450:137952. doi: 10.1016/j.cej.2022.137952. - DOI
-
- Chen J., Duan L., Sun S. Review on the development of sorbents for calcium looping. Energy Fuels. 2020;34:7806–7836. doi: 10.1021/acs.energyfuels.0c00682. - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

