Mechanism of the Photochemical Isomerization and Oxidation of 2-Butenedial: A Theoretical Study
- PMID: 37446656
- PMCID: PMC10343516
- DOI: 10.3390/molecules28134994
Mechanism of the Photochemical Isomerization and Oxidation of 2-Butenedial: A Theoretical Study
Abstract
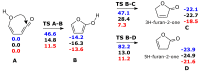
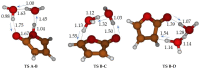
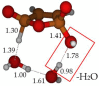
Under tropospheric conditions, 2-butenedial is photochemically removed to produce secondary organic aerosol. Upon solar irradiation in the lower troposphere, the main photochemical products are ketene-enol (a key intermediate product), furanones, and maleic anhydride. The oxidative reaction mechanism was studied using the multireference method CASSCF to explore the hypersurface of the two most accessible singlet excited states, and by DFT for the ground state. Photoisomerization of 2-butenedial in the first excited state directly produces ground state ketene-enol upon nonradiative relaxation. From this intermediate, furan-2-ol and successively 3H-furan-2-one and 5H-furan-2-one are formed. The cooperative effect of two water molecules is essential to catalyze the cyclization of ketene-enol to furan-2-ol, followed by hydrogen transfers to furanones. Two water molecules are also necessary to form maleic anhydride from furan-2-ol. For this last reaction, in which one extra oxygen must be acquired, we hypothesize a mechanism with singlet oxygen as the oxidant.
Keywords: 2-butenedial; CASSCF; CCSD(T); DFT; reaction mechanism; tropospheric oxidation.
Conflict of interest statement
The authors declare that they have no known competing financial interest or personal relationships that could have appeared to influence the work reported in this paper.
Figures
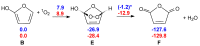
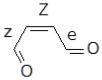
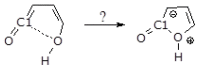
Similar articles
-
Tropospheric Photochemistry of 2-Butenedial: Role of the Triplet States, CO and Acrolein Formation, and the Experimentally Unidentified Carbonyl Compound-Theoretical Study.Molecules. 2024 Jan 24;29(3):575. doi: 10.3390/molecules29030575. Molecules. 2024. PMID: 38338321 Free PMC article.
-
Photochemistry of 2-butenedial and 4-oxo-2-pentenal under atmospheric boundary layer conditions.Phys Chem Chem Phys. 2019 Jan 21;21(3):1160-1171. doi: 10.1039/c8cp06437g. Epub 2019 Jan 8. Phys Chem Chem Phys. 2019. PMID: 30620029
-
Structural studies on cycloadducts of furan, 2-methoxyfuran, and 5-trimethylsilylcyclopentadiene with maleic anhydride and N-methylmaleimide.J Org Chem. 2008 Jan 4;73(1):151-6. doi: 10.1021/jo7018575. Epub 2007 Dec 11. J Org Chem. 2008. PMID: 18069850
-
Selective conversion of furfural to maleic anhydride and furan with VO(x)/Al(2)O(3) catalysts.ChemSusChem. 2012 Oct;5(10):1984-90. doi: 10.1002/cssc.201200167. Epub 2012 Jul 30. ChemSusChem. 2012. PMID: 22847991
-
Using singlet oxygen to synthesize polyoxygenated natural products from furans.Acc Chem Res. 2008 Aug;41(8):1001-11. doi: 10.1021/ar800023v. Epub 2008 Jul 8. Acc Chem Res. 2008. PMID: 18605738 Review.
Cited by
-
Tropospheric Photochemistry of 2-Butenedial: Role of the Triplet States, CO and Acrolein Formation, and the Experimentally Unidentified Carbonyl Compound-Theoretical Study.Molecules. 2024 Jan 24;29(3):575. doi: 10.3390/molecules29030575. Molecules. 2024. PMID: 38338321 Free PMC article.
References
-
- Obermeyer G., Aschmann S.M., Atkinson R., Arey J. Carbonyl atmospheric reaction products of aromatic hydrocarbons in ambient air. Atmos. Environ. 2009;43:3736–3744. doi: 10.1016/j.atmosenv.2009.04.015. - DOI
-
- Yokelson R.J., Karl T., Artaxo P., Blake D.R., Christian T.J., Griffith D.W.T., Guenther A., Hao W.M. The Tropical Forest and Fire Emissions Experiment: Overview and airborne fire emission factor measurements. Atmos. Chem. Phys. 2007;7:5175–5196. doi: 10.5194/acp-7-5175-2007. - DOI
-
- Andino J.M., Smith J.N., Flagan R.C., Goddard W.A., Seinfeld J.H. Mechanism of Atmospheric Photooxidation of Aromatics: A Theoretical Study. J. Phys. Chem. 1996;100:10967–10980. doi: 10.1021/jp952935l. - DOI
-
- Atkinson R. Atmospheric chemistry of VOCs and NOx. Atmos. Environ. 2000;34:2063–2101. doi: 10.1016/S1352-2310(99)00460-4. - DOI

