Hyperoside Nanomicelles Alleviate Atherosclerosis by Modulating the Lipid Profile and Intestinal Flora Structure in High-Fat-Diet-Fed Apolipoprotein-E-Deficient Mice
- PMID: 37446750
- PMCID: PMC10343736
- DOI: 10.3390/molecules28135088
Hyperoside Nanomicelles Alleviate Atherosclerosis by Modulating the Lipid Profile and Intestinal Flora Structure in High-Fat-Diet-Fed Apolipoprotein-E-Deficient Mice
Abstract
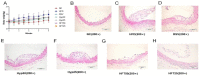
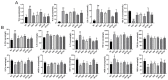
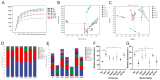
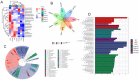
Atherosclerosis (AS) is a serious threat to human health and the main pathological basis of cardiovascular disease. Hyperoside (Hyp), a flavonoid found mainly in traditional Chinese herbs, can exert antitumor, anti-inflammatory, antioxidant, and cardiovascular-protective effects. Herein, we prepared hybrid nanomicelles (HFT) comprising Hyp loaded into pluronic F-127 and polyethylene glycol 1000 vitamin E succinate and assessed their effects on AS. To establish an AS model, apolipoprotein-E-deficient (ApoE-/-) mice were fed a high-fat diet. We then analyzed the effects of HFT on AS-induced changes in aortic tissues and metabolic markers, simultaneously assessing changes in gut flora community structure. In mice with AS, HFT significantly reduced the aortic plaque area; decreased levels of total cholesterol, triglyceride, low-density lipoprotein cholesterol, inflammatory factors, and inducible nitric oxide synthase (NOS); increased high-density lipoprotein cholesterol, endothelial NOS, superoxide dismutase, catalase, and glutathione levels; and promoted the proliferation of beneficial gut bacteria. HFT could regulate intestinal flora structure and lipid metabolism and inhibit inflammatory responses. These beneficial effects may be mediated by inhibiting nuclear factor kappa B signal activation, reducing inflammatory factor expression and improving gut microflora structure and dyslipidemia. The present study provides an empirical basis for the development and clinical application of new dosage forms of Hyp.
Keywords: apolipoprotein E; atherosclerosis; hyperoside nanomicelle; intestinal flora; serum lipidomics.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Karunakaran D., Nguyen M.A., Geoffrion M., Vreeken D., Lister Z., Cheng H.S., Otte N., Essebier P., Wyatt H., Kandiah J.W., et al. RIPK1 Expression Associates with Inflammation in Early Atherosclerosis in Humans and Can Be Therapeutically Silenced to Reduce NF-κB Activation and Atherogenesis in Mice. Circulation. 2021;143:163–177. doi: 10.1161/CIRCULATIONAHA.118.038379. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

