Antibody production and tolerance to the α-gal epitope as models for understanding and preventing the immune response to incompatible ABO carbohydrate antigens and for α-gal therapies
- PMID: 37449060
- PMCID: PMC10338101
- DOI: 10.3389/fmolb.2023.1209974
Antibody production and tolerance to the α-gal epitope as models for understanding and preventing the immune response to incompatible ABO carbohydrate antigens and for α-gal therapies
Abstract
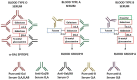
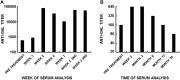
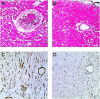
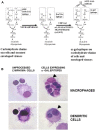
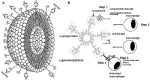
This review describes the significance of the α-gal epitope (Galα-3Galβ1-4GlcNAc-R) as the core of human blood-group A and B antigens (A and B antigens), determines in mouse models the principles underlying the immune response to these antigens, and suggests future strategies for the induction of immune tolerance to incompatible A and B antigens in human allografts. Carbohydrate antigens, such as ABO antigens and the α-gal epitope, differ from protein antigens in that they do not interact with T cells, but B cells interacting with them require T-cell help for their activation. The α-gal epitope is the core of both A and B antigens and is the ligand of the natural anti-Gal antibody, which is abundant in all humans. In A and O individuals, anti-Gal clones (called anti-Gal/B) comprise >85% of the so-called anti-B activity and bind to the B antigen in facets that do not include fucose-linked α1-2 to the core α-gal. As many as 1% of B cells are anti-Gal B cells. Activation of quiescent anti-Gal B cells upon exposure to α-gal epitopes on xenografts and some protozoa can increase the titer of anti-Gal by 100-fold. α1,3-Galactosyltransferase knockout (GT-KO) mice lack α-gal epitopes and can produce anti-Gal. These mice simulate human recipients of ABO-incompatible human allografts. Exposure for 2-4 weeks of naïve and memory mouse anti-Gal B cells to α-gal epitopes in the heterotopically grafted wild-type (WT) mouse heart results in the elimination of these cells and immune tolerance to this epitope. Shorter exposures of 7 days of anti-Gal B cells to α-gal epitopes in the WT heart result in the production of accommodating anti-Gal antibodies that bind to α-gal epitopes but do not lyse cells or reject the graft. Tolerance to α-gal epitopes due to the elimination of naïve and memory anti-Gal B cells can be further induced by 2 weeks in vivo exposure to WT lymphocytes or autologous lymphocytes engineered to present α-gal epitopes by transduction of the α1,3-galactosyltransferase gene. These mouse studies suggest that autologous human lymphocytes similarly engineered to present the A or B antigen may induce corresponding tolerance in recipients of ABO-incompatible allografts. The review further summarizes experimental works demonstrating the efficacy of α-gal therapies in amplifying anti-viral and anti-tumor immune-protection and regeneration of injured tissues.
Keywords: ABO-incompatible antigens; alpha-gal epitope; alpha-gal nanoparticles; alpha-gal therapies; anti-Gal; immune accommodation; immune tolerance.
Copyright © 2023 Galili.
Conflict of interest statement
The author declares that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures





Similar articles
-
Xenotransplantation and ABO incompatible transplantation: the similarities they share.Transfus Apher Sci. 2006 Aug;35(1):45-58. doi: 10.1016/j.transci.2006.05.007. Epub 2006 Aug 14. Transfus Apher Sci. 2006. PMID: 16905361 Review.
-
Mouse-heart grafts expressing an incompatible carbohydrate antigen. II. Transition from accommodation to tolerance.Transplantation. 2004 Feb 15;77(3):366-73. doi: 10.1097/01.TP.0000109276.57772.6D. Transplantation. 2004. PMID: 14966409
-
Immune response, accommodation, and tolerance to transplantation carbohydrate antigens.Transplantation. 2004 Oct 27;78(8):1093-8. doi: 10.1097/01.tp.0000142673.32394.95. Transplantation. 2004. PMID: 15502700 Review.
-
Anti-gal A/B, a novel anti-blood group antibody identified in recipients of abo-incompatible kidney allografts.Transplantation. 2002 Dec 15;74(11):1574-80. doi: 10.1097/00007890-200212150-00015. Transplantation. 2002. PMID: 12490791
-
Induction of immune tolerance to a transplantation carbohydrate antigen by gene therapy with autologous lymphocytes transduced with adenovirus containing the corresponding glycosyltransferase gene.Gene Ther. 2004 Feb;11(3):292-301. doi: 10.1038/sj.gt.3302178. Gene Ther. 2004. PMID: 14737089
Cited by
-
A systematic review reveals conflicting evidence for the prevalence of antibodies against the sialic acid 'xenoautoantigen' Neu5Gc in humans and the need for a standardised approach to quantification.Front Mol Biosci. 2024 Apr 26;11:1390711. doi: 10.3389/fmolb.2024.1390711. eCollection 2024. Front Mol Biosci. 2024. PMID: 38737334 Free PMC article. Review.
-
α-Gal Nanoparticles in CNS Trauma: I. In Vitro Activation of Microglia Towards a Pro-Healing State.Tissue Eng Regen Med. 2024 Apr;21(3):409-419. doi: 10.1007/s13770-023-00613-1. Epub 2023 Dec 15. Tissue Eng Regen Med. 2024. PMID: 38099990 Free PMC article.
-
α-Gal Nanoparticles in CNS Trauma: II. Immunomodulation Following Spinal Cord Injury (SCI) Improves Functional Outcomes.Tissue Eng Regen Med. 2024 Apr;21(3):437-453. doi: 10.1007/s13770-023-00616-y. Epub 2024 Feb 3. Tissue Eng Regen Med. 2024. PMID: 38308742 Free PMC article.
References
-
- Alexandre G. P., Squifflet J. P., DeBruyere B. M., Latinne D., Reding R., Gianello P., et al. (1987). Present experiences in a series of 26 ABO-incompatible living donor renal allografts. Transpl. Proc. 19, 4538–4542. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

