Case report: Use of therapeutic drug monitoring and pharmacogenetic testing as opportunities to individualize care in a case of flecainide toxicity after fetal supraventricular tachycardia
- PMID: 37449265
- PMCID: PMC10337585
- DOI: 10.3389/fped.2023.1168619
Case report: Use of therapeutic drug monitoring and pharmacogenetic testing as opportunities to individualize care in a case of flecainide toxicity after fetal supraventricular tachycardia
Abstract
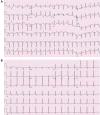
Flecainide is a class IC antiarrhythmic utilized in prophylaxis of refractory paroxysmal supraventricular tachycardias in pediatric populations. Despite being a highly effective agent, its narrow therapeutic index increases the risk of toxicity and proarrhythmic events, including wide-complex tachycardia. In the absence of direct plasma sampling in the fetus to quantitate flecainide systemic concentrations, clinicians typically make drug dosing decisions from maternal plasma concentrations and QRS duration on maternal ECGs. There remains a paucity of standard guidelines and data to inform the timing and frequency of the aforementioned test in pregnancy and timing of flecainide discontinuation prior to childbirth. Flecainide primarily undergoes metabolism via cytochrome P450 (CYP). Given the variance of CYP-mediated metabolism at the level of the individual patient, pharmacogenomics can be considered in patients who present with flecainide toxicity to determine the maternal vs. fetal factors as an etiology for the event. Finally, pharmacogenetic testing can be utilized as an adjunct to guide flecainide dosing decisions, but must be done with caution in neonates <2 weeks of age. This case report highlights utilization of pharmacogenomic testing and therapeutic drug monitoring as adjuncts to guide therapy for a newborn with refractory supraventricular tachycardia, who experienced flecainide toxicity immediately post-partum and was trialed unsuccessfully on multiple alternative antiarrhythmics without rhythm control.
Keywords: drug monitoring; flecainide; pharmacogenetic; pharmacogenomics; toxicity.
© 2023 Palmen, Sandritter, Malloy-Walton, Follansbee and Wagner.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Flecainide intoxication in pediatric patients with supraventricular tachycardia.Ann Pediatr Cardiol. 2020 Jul-Sep;13(3):264-266. doi: 10.4103/apc.APC_116_19. Epub 2020 Jun 29. Ann Pediatr Cardiol. 2020. PMID: 32863668 Free PMC article.
-
Correlation of maternal flecainide concentrations and therapeutic effect in fetal supraventricular tachycardia.Heart Rhythm. 2014 Nov;11(11):2047-53. doi: 10.1016/j.hrthm.2014.07.031. Epub 2014 Jul 25. Heart Rhythm. 2014. PMID: 25068572
-
Fetal and maternal magnetocardiography during flecainide therapy for supraventricular tachycardia.Obstet Gynecol. 2006 Sep;108(3 Pt 2):767-71. doi: 10.1097/01.AOG.0000197063.25177.69. Obstet Gynecol. 2006. PMID: 17018496
-
Pharmacologic management of supraventricular tachycardias in children. Part 1: Wolff-Parkinson-White and atrioventricular nodal reentry.Ann Pharmacother. 1997 Oct;31(10):1227-43. doi: 10.1177/106002809703101016. Ann Pharmacother. 1997. PMID: 9337449 Review.
-
Pharmacologic management of supraventricular tachycardias in children. Part 2: Atrial flutter, atrial fibrillation, and junctional and atrial ectopic tachycardia.Ann Pharmacother. 1997 Nov;31(11):1347-59. doi: 10.1177/106002809703101113. Ann Pharmacother. 1997. PMID: 9391691 Review.
Cited by
-
Pediatric flecainide pharmacogenomics: a roadmap to delivering precision-based care to pediatrics arrhythmias.Front Pharmacol. 2024 Dec 16;15:1477485. doi: 10.3389/fphar.2024.1477485. eCollection 2024. Front Pharmacol. 2024. PMID: 39741635 Free PMC article. Review.
References
Publication types
LinkOut - more resources
Full Text Sources

