Hernia Prevention Using Biologic Mesh and/or Small Bites: A Multispecialty 2 × 2 Factorial Randomized Controlled Trial
- PMID: 37458369
- PMCID: PMC10574223
- DOI: 10.1097/XCS.0000000000000705
Hernia Prevention Using Biologic Mesh and/or Small Bites: A Multispecialty 2 × 2 Factorial Randomized Controlled Trial
Abstract
Background: Ventral incisional hernias are the most common complication after abdominal operation. Randomized trials have shown efficacy of prophylactic synthetic mesh and small bites. Adoption of these practices has been limited due to concerns with placement of synthetic mesh in contaminated cases and small bites in an overweight population. We sought to assess the efficacy of prophylactic biologic mesh and small bites to prevent postoperative major complications: ventral incisional hernias, surgical site infection, reoperation, and death.
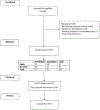
Study design: High-risk patients (overweight/obese, current smoker) undergoing abdominal operation with a midline incision (5 cm or greater) were randomized (2 × 2 factorial trial) to receive either sublay biologic mesh or no mesh and either small bites (0.5 × 0. 5cm) or large bites (1 × 1 cm) fascial closure. The primary outcome measure was major complications at 1 year postoperative. CONSORT guidelines were followed, and this study was registered on clinicaltrials.gov (NCT03148496). Assuming α = 0.05, β = 0.20, and Δ = 20%, it was estimated that 105 patients were needed. Primary outcome was assessed using Fisher's exact test.
Results: A total of 107 patients were randomized: 52 (49%) to mesh, 55 (51%) to no mesh, 55 (51%) to small bites, and 52 (49%) to large bites. Of the patients, 16% were smokers, 31% were overweight, and 55% were obese. At 1 year postoperative, there were no differences in major complications between groups (mesh vs no mesh 21% vs 16%, p = 0.62; small vs large bites 18% vs 19%, p = 1.00).
Conclusions: In this trial, biologic mesh and small bites appear to have no benefit. Further randomized trials are needed among high-risk patients before widespread adoption of prophylactic biologic mesh or small bites.
Copyright © 2023 by the American College of Surgeons. Published by Wolters Kluwer Health, Inc. All rights reserved.
Figures
Comment in
-
Invited Commentary: Is the Biological Clock for Biologic Mesh in Abdominal Surgery Nearing Its End?J Am Coll Surg. 2023 Aug 1;237(2):317-318. doi: 10.1097/XCS.0000000000000717. Epub 2023 Apr 14. J Am Coll Surg. 2023. PMID: 37057831 No abstract available.
Similar articles
-
Prophylactic Mesh Reinforcement Versus Primary Suture for Abdominal Wall Closure after Elective Abdominal Aortic Aneurysm Repair with Midline Laparotomy Incision: Updated Systematic Review Including Time-To-Event Meta-Analysis and Trial Sequential Analysis of Randomized Controlled Trials.Ann Vasc Surg. 2024 Dec;109:149-161. doi: 10.1016/j.avsg.2024.06.026. Epub 2024 Jul 16. Ann Vasc Surg. 2024. PMID: 39025216
-
Closure methods for laparotomy incisions for preventing incisional hernias and other wound complications.Cochrane Database Syst Rev. 2017 Nov 3;11(11):CD005661. doi: 10.1002/14651858.CD005661.pub2. Cochrane Database Syst Rev. 2017. PMID: 29099149 Free PMC article.
-
Prescription of Controlled Substances: Benefits and Risks.2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2025 Jul 6. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 30726003 Free Books & Documents.
-
Mesh versus non-mesh for inguinal and femoral hernia repair.Cochrane Database Syst Rev. 2018 Sep 13;9(9):CD011517. doi: 10.1002/14651858.CD011517.pub2. Cochrane Database Syst Rev. 2018. PMID: 30209805 Free PMC article.
-
Mesh-reinforced compared with primary closure in emergent laparotomies for incarcerated ventral incisional hernias: Is the added complexity justified?Surgery. 2025 Sep;185:109536. doi: 10.1016/j.surg.2025.109536. Epub 2025 Jul 10. Surgery. 2025. PMID: 40644740
References
-
- Olavarria OA, Dhanani NH, Bernardi K, et al. Prophylactic mesh reinforcement for prevention of midline incisional hernias: a publication bias adjusted meta-analysis. Ann Surg 2023;277:e162–e169. - PubMed
-
- Jairam AP, Timmermans L, Eker HH, et al.; PRIMA Trialist Group. Prevention of incisional hernia with prophylactic onlay and sublay mesh reinforcement versus primary suture only in midline laparotomies (PRIMA): 2-year follow-up of a multicentre, double-blind, randomised controlled trial. Lancet 2017;390:567–576. - PubMed
-
- Glauser PM, Brosi P, Speich B, et al. Prophylactic intraperitoneal onlay mesh following midline laparotomy-long-term results of a randomized controlled trial. World J Surg 2019;43:1669–1675. - PubMed
-
- Millbourn D, Cengiz Y, Israelsson LA. Effect of stitch length on wound complications after closure of midline incisions: a randomized controlled trial. Arch Surg 2009;144:1056–1059. - PubMed
-
- Deerenberg EB, Harlaar JJ, Steyerberg EW, et al. Small bites versus large bites for closure of abdominal midline incisions (STITCH): a double-blind, multicentre, randomised controlled trial. Lancet 2015;386:1254–1260. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical