Utility of Minimal Physiologically Based Pharmacokinetic Models for Assessing Fractional Distribution, Oral Absorption, and Series-Compartment Models of Hepatic Clearance
- PMID: 37460222
- PMCID: PMC10506700
- DOI: 10.1124/dmd.123.001403
Utility of Minimal Physiologically Based Pharmacokinetic Models for Assessing Fractional Distribution, Oral Absorption, and Series-Compartment Models of Hepatic Clearance
Abstract
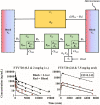
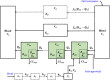
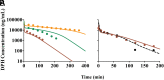
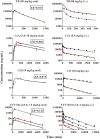
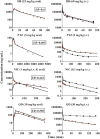
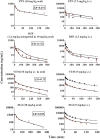
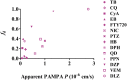
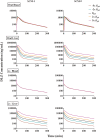
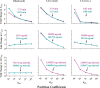
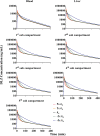
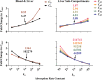
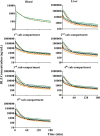
Minimal physiologically based pharmacokinetic (mPBPK) models are physiologically relevant, require less information than full PBPK models, and offer flexibility in pharmacokinetics (PK). The well-stirred hepatic model (WSM) is commonly used in PBPK, whereas the more plausible dispersion model (DM) poses computational complexities. The series-compartment model (SCM) mimics the DM but is easier to operate. This work implements the SCM and mPBPK models for assessing fractional tissue distribution, oral absorption, and hepatic clearance using literature-reported blood and liver concentration-time data in rats for compounds mainly cleared by the liver. Further handled were various complexities, including nonlinear hepatic binding and metabolism, differing absorption kinetics, and sites of administration. The SCM containing one to five (n) liver subcompartments yields similar fittings and provides comparable estimates for hepatic extraction ratio (ER), prehepatic availability (Fg ), and first-order absorption rate constants (ka ). However, they produce decreased intrinsic clearances (CLint ) and liver-to-plasma partition coefficients (Kph ) with increasing n as expected. Model simulations demonstrated changes in intravenous and oral PK profiles with alterations in Kph and ka and with hepatic metabolic zonation. The permeability (PAMPA P) of the various compounds well explained the fitted fractional distribution (fd ) parameters. The SCM and mPBPK models offer advantages in distinguishing systemic, extrahepatic, and hepatic clearances. The SCM allows for incorporation of liver zonation and is useful in assessing changes in internal concentration gradients potentially masked by similar blood PK profiles. Improved assessment of intraorgan drug concentrations may offer insights into active moieties driving metabolism, biliary excretion, pharmacodynamics, and hepatic toxicity. SIGNIFICANCE STATEMENT: The minimal physiologically based pharmacokinetic model and the series-compartment model are useful in assessing oral absorption and hepatic clearance. They add flexibility in accounting for various drug- or system-specific complexities, including fractional distribution, nonlinear binding and saturable hepatic metabolism, and hepatic zonation. These models can offer improved insights into the intraorgan concentrations that reflect physiologically active moieties often driving disposition, pharmacodynamics, and toxicity.
Copyright © 2023 by The American Society for Pharmacology and Experimental Therapeutics.
Figures


References
-
- Abu-Zahra TN, Pang KS (2000) Effect of zonal transport and metabolism on hepatic removal: enalapril hydrolysis in zonal, isolated rat hepatocytes in vitro and correlation with perfusion data. Drug Metab Dispos 28:807–813. - PubMed
-
- Adelusi SA, Salako LA (1982) Kinetics of the distribution and elimination of chloroquine in the rat. Gen Pharmacol 13:433–437. - PubMed
-
- Allison AC, Young MR (1964) Uptake of dyes and drugs by living cells in culture. Life Sci 3:1407–1414. - PubMed
-
- Anissimov YG, Roberts MS (2002) A compartmental model of hepatic disposition kinetics: 1. Model development and application to linear kinetics. J Pharmacokinet Pharmacodyn 29:131–156. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

