Glutathione trisulfide prevents lipopolysaccharide-induced retinal inflammation via inhibition of proinflammatory cytokine production in glial cells
- PMID: 37460786
- PMCID: PMC10352383
- DOI: 10.1038/s41598-023-38696-4
Glutathione trisulfide prevents lipopolysaccharide-induced retinal inflammation via inhibition of proinflammatory cytokine production in glial cells
Abstract
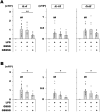
We aimed to investigate the impact of glutathione trisulfide (GSSSG) on lipopolysaccharide (LPS)-induced inflammation in retinal glia. Inflammatory responses in mouse-derived glial cells and Wistar rat retinas were stimulated with administration of LPS. Cell survival and proinflammatory cytokine production were examined using the Calcein-AM assay, and reverse transcription-quantitative polymerase chain reaction (RT-qPCR) and enzyme-linked immunosorbent assay (ELISA), respectively. Retinal microglia were visualized with immunohistochemistry for Iba1. Administration of LPS (10 µg/mL) or GSSSG (less than 100 µM) did not affect survival of cultured primary Müller cells and established microglial cells (BV-2). RT-qPCR and ELISA indicated that GSSSG inhibited LPS-induced gene upregulation and protein secretion of proinflammatory cytokines in these glial cells and rat retinas. GSSSG inhibited LPS-induced activation of TGF-β-activated kinase 1 (TAK1), which is an upstream kinase of NF-κB, in BV-2 cells. Finally, in vivo experiments indicated that intravitreal administration of GSSSG but not its relative glutathione disulfide (GSSG) inhibited LPS (500 ng)-induced accumulation of Iba1-immunopositive microglia in rat retinas. Taken together, GSSSG has the potential to prevent pathogenesis of inflammation-associated ocular diseases by inhibiting proinflammatory cytokine expression in retinal glial cells.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures





References
-
- Le J, Vilcek J. Interleukin 6: A multifunctional cytokine regulating immune reactions and the acute phase protein response. Lab. Invest. 1989;61:588–602. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

