Achieving comparability in glycemic control between antidiabetic treatment strategies in pregnancy when using real world data
- PMID: 37461243
- PMCID: PMC10792121
- DOI: 10.1002/pds.5665
Achieving comparability in glycemic control between antidiabetic treatment strategies in pregnancy when using real world data
Abstract
Purpose: Healthcare utilization databases often lack information on glycemic control, a key confounder when studying the safety of antidiabetic treatments, since patients with worse control are channeled to second-line agents, in particular insulin, versus first-line agents such as metformin. We evaluated whether adjustment for measured characteristics attains balance in glycemic control when comparing antidiabetic treatment strategies in pregnant women with pregestational type 2 diabetes (T2DM).
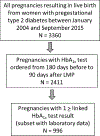
Methods: In a US insurance claims database, we identified 3360 women with T2DM pregnant between 2004 and 2015, of whom a subset of 996 had data on hemoglobin A1c (HbA1c ) levels. We selected insulin only as the comparator group and used propensity score (PS)-matching on comorbidities and proxies of diabetes severity, but not on HbA1c , to adjust for confounding. We used standardized differences (st.diff) to assess balance in claims-based covariates and mean HbA1c (% ± SD) in the subset.
Results: There were imbalances in claims-based covariates before PS-matching, with smaller differences when both treatment strategies included insulin. After PS-matching, balance was achieved in most claims-based covariates (st.diff <0.1). Mean HbA1c was similar before and after PS-matching when both treatments included insulin (e.g., 7.1 ± 1.5 vs. 7.7 ± 1.8 and 7.1 ± 1.5 vs. 7.5 ± 1.7, respectively, for metformin + insulin vs. insulin only). Differences in mean HbA1c remained after PS-matching when non-insulin treatments were compared to treatments including insulin (e.g., 6.3 ± 1.1 vs. 7.6 ± 1.7 for metformin only vs. insulin only).
Conclusions: Balance in both claims-based characteristics and glycemic control was attained after restricting the population to women with T2DM and comparing treatment strategies indicated for patients with similar diabetes severity. When comparing treatment strategies with versus without insulin, differences in glycemic control persisted after PS-matching even when balance was attained for other measured characteristics.
Keywords: antidiabetic medication; channeling bias; confounding; glycemic control; pregnancy; real world data; type 2 diabetes mellitus.
© 2023 The Authors. Pharmacoepidemiology and Drug Safety published by John Wiley & Sons Ltd.
Conflict of interest statement
Figures


Similar articles
-
Claims-based studies of oral glucose-lowering medications can achieve balance in critical clinical variables only observed in electronic health records.Diabetes Obes Metab. 2018 Apr;20(4):974-984. doi: 10.1111/dom.13184. Epub 2018 Jan 12. Diabetes Obes Metab. 2018. PMID: 29206336 Free PMC article.
-
Effectiveness of the Lilly Connected Care Program in Improving Glycemic Management Among Patients With Type 2 Diabetes in China: Retrospective Real-world Study.J Med Internet Res. 2023 Apr 25;25:e38680. doi: 10.2196/38680. J Med Internet Res. 2023. PMID: 37097724 Free PMC article.
-
Real world management of pregestational diabetes not achieving glycemic control for many patients in the UK.Pharmacoepidemiol Drug Saf. 2018 Aug;27(8):940-948. doi: 10.1002/pds.4553. Epub 2018 May 8. Pharmacoepidemiol Drug Saf. 2018. PMID: 29740916
-
Repaglinide : a pharmacoeconomic review of its use in type 2 diabetes mellitus.Pharmacoeconomics. 2004;22(6):389-411. doi: 10.2165/00019053-200422060-00005. Pharmacoeconomics. 2004. PMID: 15099124 Review.
-
Advancing therapy in type 2 diabetes mellitus with early, comprehensive progression from oral agents to insulin therapy.Clin Ther. 2007;29 Spec No:1236-53. Clin Ther. 2007. PMID: 18046925 Review.
Cited by
-
Perinatal Outcomes Associated With Metformin Use During Pregnancy in Women With Pregestational Type 2 Diabetes Mellitus.Diabetes Care. 2024 Sep 1;47(9):1688-1695. doi: 10.2337/dc23-2056. Diabetes Care. 2024. PMID: 39042587
-
Safety of GLP-1 Receptor Agonists and Other Second-Line Antidiabetics in Early Pregnancy.JAMA Intern Med. 2024 Feb 1;184(2):144-152. doi: 10.1001/jamainternmed.2023.6663. JAMA Intern Med. 2024. PMID: 38079178 Free PMC article.
References
-
- Cho NH, Shaw JE, Karuranga S, Huang Y, da Rocha Fernandes JD, Ohlrogge AW, et al. IDF Diabetes Atlas: Global estimates of diabetes prevalence for 2017 and projections for 2045. Diabetes Res Clin Pract. 2018;138:271–81. - PubMed
-
- American Diabetes Association. 9. Pharmacologic Approaches to Glycemic Treatment: Standards of Medical Care in Diabetes-2021. Diabetes care. 2021;44(Suppl 1):S111–s24. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

