Histone deacetylase III interactions with BK polyomavirus large tumor antigen may affect protein stability
- PMID: 37464367
- PMCID: PMC10354968
- DOI: 10.1186/s12985-023-02128-6
Histone deacetylase III interactions with BK polyomavirus large tumor antigen may affect protein stability
Abstract
Background: Human polyomavirus BK (BKPyV) causes associated nephropathy and contributes to urinary tract cancer development in renal transplant recipients. Large tumor antigen (LT) is an early protein essential in the polyomavirus life cycle. Protein acetylation plays a critical role in regulating protein stability, so this study investigated the acetylation of the BKPyV LT protein.
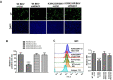
Methods: The BKPyV LT nucleotide was synthesized, and the protein was expressed by transfection into permissive cells. The BKPyV LT protein was immunoprecipitated and subjected to LC-MS/MS analysis to determine the acetylation residues. The relative lysine was then mutated to arginine in the LT nucleotide and BKPyV genome to analyze the role of LT lysine acetylation in the BKPyV life cycle.
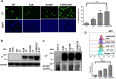
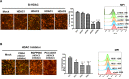
Results: BKPyV LT acetylation sites were identified at Lys3 and Lys230 by mass spectrometry. HDAC3 and HDAC8 and their deacetylation activity are required for BKPyV LT expression. In addition, mutations of Lys3 and Lys230 to arginine increased LT expression, and the interaction of HDAC3 and LT was confirmed by coimmunoprecipitation.
Conclusions: HDAC3 is a newly identified protein that interacts with BKPyV LT, and LT acetylation plays a vital role in the BKPyV life cycle.
Keywords: Acetylation; BKPyV; HDAC3; Large T antigen; Protein stability.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures


References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

