Inflammasome activation and pyroptosis mediate coagulopathy and inflammation in Salmonella systemic infection
- PMID: 37467711
- PMCID: PMC10693354
- DOI: 10.1016/j.micres.2023.127460
Inflammasome activation and pyroptosis mediate coagulopathy and inflammation in Salmonella systemic infection
Abstract
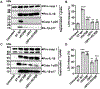
Inflammasome activation is a critical defense mechanism against bacterial infection. Previous studies suggest that inflammasome activation protects against Salmonella oral infection. Here we find inflammasome activation plays a critical role in the pathogenesis of Salmonella systemic infection. We show that in a systemic infection model by i.p. injection of Salmonella, deficiency of caspase-1 or gasdermin-D prolonged survival time, reduced plasma concentrations of the proinflammatory cytokines IL-1β, IL-6 and TNFα. These deficiencies also protected against coagulopathy during Salmonella infection as evidenced by diminished prolongation of prothrombin time and increase in plasma thrombin-antithrombin complex concentrations in the caspase-1 or gasdermin-D deficient mice. Activation of the NAIP/NLRC4 inflammasome by flagellin and/or the components of the SPI1 type 3 secretion system played a critical role in Salmonella-induced coagulopathy. In the absence of flagellin and SPI1, the Salmonella mutant strain still triggered coagulopathy through the caspase-11/NLRP3 pathway. Our results reveal a previously undisclosed role of the inflammasomes and pyroptosis in the pathogenesis of Salmonella systemic infection.
Keywords: Coagulation; DIC; Inflammasome; Macrophage; Pyroptosis; Salmonella.
Copyright © 2023 Elsevier GmbH. All rights reserved.
Conflict of interest statement
Declaration of Competing Interest The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures
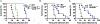
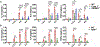
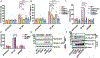

References
-
- Appiah GD, Mpimbaza A, Lamorde M, Freeman M, Kajumbula H, Salah Z, Kugeler K, Mikoleit M, White PB, Kapisi J, Borchert J, Sserwanga A, Van Dyne S, Mead P, Kim S, Lauer AC, Winstead A, Manabe YC, Flick RJ, Mintz E, 2021. Salmonella bloodstream infections in hospitalized children with acute febrile illness-Uganda, 2016–2019. Am. J. Trop. Med. Hyg. 105, 37–46. - PMC - PubMed
-
- Brennan MA, Cookson BT, 2000. Salmonella induces macrophage death by caspase-1-dependent necrosis. Mol. Microbiol. 38, 31–40. - PubMed
-
- Brinkmann V, Reichard U, Goosmann C, Fauler B, Uhlemann Y, Weiss DS, Weinrauch Y, Zychlinsky A, 2004. Neutrophil extracellular traps kill bacteria. Science 303, 1532–1535. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

