Diffusion-free valve for preprogrammed immunoassay with capillary microfluidics
- PMID: 37469685
- PMCID: PMC10352302
- DOI: 10.1038/s41378-023-00568-2
Diffusion-free valve for preprogrammed immunoassay with capillary microfluidics
Abstract
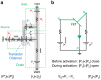
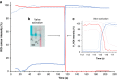
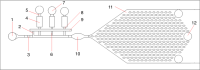
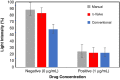
By manipulating the geometry and surface chemistry of microfluidic channels, capillary-driven microfluidics can move and stop fluids spontaneously without external instrumentation. Furthermore, complex microfluidic circuits can be preprogrammed by synchronizing the capillary pressures and encoding the surface tensions of microfluidic chips. A key component of these systems is the capillary valve. However, the main concern for these valves is the presence of unwanted diffusion during the valve loading and activation steps that can cause cross-contamination. In this study, we design and validate a novel diffusion-free capillary valve: the π-valve. This valve consists of a 3D structure and a void area. The void acts as a spacer between two fluids to avoid direct contact. When the valve is triggered, the air trapped within the void is displaced by pneumatic suction induced from the capillary flow downstream without introducing a gas bubble into the circuit. The proposed design eliminates diffusive mixing before valve activation. Numerical simulation is used to study the function and optimize the dimensions of the π-valve, and 3D printing is used to fabricate either the mould or the microfluidic chip. A comparison with a conventional valve (based on a constriction-expansion valve) demonstrates that the π-valve eliminates possible backflow into the valve and reduces the mixing and diffusion during the loading and trigger steps. As a proof-of-concept, this valve is successfully implemented in a capillary-driven circuit for the determination of benzodiazepine, achieving the successive release of 3 solutions in a 3D-printed microfluidic chip without external instrumentation. The results show a 40% increase in the fluorescence intensity using the π-valve relative to the conventional value. Overall, the π-valve prevents cross-contamination, minimizes sample use, and facilitates a sophisticated preprogrammed release of fluids, offering a promising tool for conducting automated immunoassays applicable at point-of-care testing.
Keywords: Chemistry; Electrical and electronic engineering.
© The Author(s) 2023.
Conflict of interest statement
Conflict of interestThe authors declare no competing interests.
Figures




Similar articles
-
The dynamics of capillary flow in an open-channel system featuring trigger valves.Sci Rep. 2024 Dec 30;14(1):31732. doi: 10.1038/s41598-024-82329-3. Sci Rep. 2024. PMID: 39738276 Free PMC article.
-
The dynamics of capillary flow in an open-channel system featuring trigger valves.bioRxiv [Preprint]. 2024 Nov 30:2024.09.17.613325. doi: 10.1101/2024.09.17.613325. bioRxiv. 2024. Update in: Sci Rep. 2024 Dec 30;14(1):31732. doi: 10.1038/s41598-024-82329-3. PMID: 39345588 Free PMC article. Updated. Preprint.
-
Coupling Capillary-Driven Microfluidics with Lateral Flow Immunoassay for Signal Enhancement.Biosensors (Basel). 2023 Aug 21;13(8):832. doi: 10.3390/bios13080832. Biosensors (Basel). 2023. PMID: 37622918 Free PMC article.
-
Capillary-driven microfluidics: impacts of 3D manufacturing on bioanalytical devices.Analyst. 2023 Jun 12;148(12):2657-2675. doi: 10.1039/d3an00115f. Analyst. 2023. PMID: 37166188 Review.
-
Capillary microfluidics in microchannels: from microfluidic networks to capillaric circuits.Lab Chip. 2018 Aug 7;18(16):2323-2347. doi: 10.1039/c8lc00458g. Lab Chip. 2018. PMID: 30010168 Review.
Cited by
-
Diagnostic technologies for neuroblastoma.Lab Chip. 2025 Jul 23;25(15):3630-3664. doi: 10.1039/d4lc00005f. Lab Chip. 2025. PMID: 40653949 Free PMC article. Review.
-
Wicking pumps for microfluidics.Biomicrofluidics. 2024 Nov 1;18(6):061501. doi: 10.1063/5.0218030. eCollection 2024 Dec. Biomicrofluidics. 2024. PMID: 39494378 Free PMC article. Review.
-
The dynamics of capillary flow in an open-channel system featuring trigger valves.Sci Rep. 2024 Dec 30;14(1):31732. doi: 10.1038/s41598-024-82329-3. Sci Rep. 2024. PMID: 39738276 Free PMC article.
-
The dynamics of capillary flow in an open-channel system featuring trigger valves.bioRxiv [Preprint]. 2024 Nov 30:2024.09.17.613325. doi: 10.1101/2024.09.17.613325. bioRxiv. 2024. Update in: Sci Rep. 2024 Dec 30;14(1):31732. doi: 10.1038/s41598-024-82329-3. PMID: 39345588 Free PMC article. Updated. Preprint.
-
Coupling Capillary-Driven Microfluidics with Lateral Flow Immunoassay for Signal Enhancement.Biosensors (Basel). 2023 Aug 21;13(8):832. doi: 10.3390/bios13080832. Biosensors (Basel). 2023. PMID: 37622918 Free PMC article.
References
-
- Bernetski KA, Burkhart CT, Maki KL, Schertzer MJ. Characterization of electrowetting, contact angle hysteresis, and adhesion on digital microfluidic devices with inkjet-printed electrodes. Microfluid Nanofluid. 2018;22:96. doi: 10.1007/s10404-018-2119-4. - DOI
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
