Postsynaptic Calcium Extrusion at the Mouse Neuromuscular Junction Alkalinizes the Synaptic Cleft
- PMID: 37474311
- PMCID: PMC10423045
- DOI: 10.1523/JNEUROSCI.0815-23.2023
Postsynaptic Calcium Extrusion at the Mouse Neuromuscular Junction Alkalinizes the Synaptic Cleft
Abstract
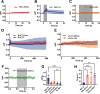
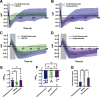
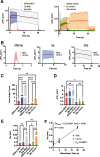
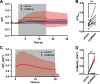
Neurotransmission is shaped by extracellular pH. Alkalization enhances pH-sensitive transmitter release and receptor activation, whereas acidification inhibits these processes and can activate acid-sensitive conductances in the synaptic cleft. Previous work has shown that the synaptic cleft can either acidify because of synaptic vesicular release and/or alkalize because of Ca2+ extrusion by the plasma membrane ATPase (PMCA). The direction of change differs across synapse types. At the mammalian neuromuscular junction (NMJ), the direction and magnitude of pH transients in the synaptic cleft during transmission remain ambiguous. We set out to elucidate the extracellular pH transients that occur at this cholinergic synapse under near-physiological conditions and identify their sources. We monitored pH-dependent changes in the synaptic cleft of the mouse levator auris longus using viral expression of the pseudoratiometric probe pHusion-Ex in the muscle. Using mice from both sexes, a significant and prolonged alkalization occurred when stimulating the connected nerve for 5 s at 50 Hz, which was dependent on postsynaptic intracellular Ca2+ release. Sustained stimulation for a longer duration (20 s at 50 Hz) caused additional prolonged net acidification at the cleft. To investigate the mechanism underlying cleft alkalization, we used muscle-expressed GCaMP3 to monitor the contribution of postsynaptic Ca2+ Activity-induced liberation of intracellular Ca2+ in muscle positively correlated with alkalization of the synaptic cleft, whereas inhibiting PMCA significantly decreased the extent of cleft alkalization. Thus, cholinergic synapses of the mouse NMJ typically alkalize because of cytosolic Ca2+ liberated in muscle during activity, unless under highly strenuous conditions where acidification predominates.SIGNIFICANCE STATEMENT Changes in synaptic cleft pH alter neurotransmission, acting on receptors and channels on both sides of the synapse. Synaptic acidification has been associated with a myriad of diseases in the central and peripheral nervous system. Here, we report that in near-physiological recording conditions the cholinergic neuromuscular junction shows use-dependent bidirectional changes in synaptic cleft pH-immediate alkalinization and a long-lasting acidification under prolonged stimulation. These results provide further insight into physiologically relevant changes at cholinergic synapses that have not been defined previously. Understanding and identifying synaptic pH transients during and after neuronal activity provides insight into short-term synaptic plasticity synapses and may identify therapeutic targets for diseases.
Keywords: acetylcholine; mouse; neuromuscular junction; pH; synapse; synaptic transmission.
Copyright © 2023 Durbin et al.
Figures


References
-
- Alijevic O, Bignucolo O, Hichri E, Peng Z, Kucera JP, Kellenberger S (2020) Slowing of the time course of acidification decreases the acid-sensing ion channel 1a current amplitude and modulates action potential firing in neurons. Front Cell Neurosci 14:41. 10.3389/fncel.2020.00041 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
