A unique maternal and placental galectin signature upon SARS-CoV-2 infection suggests galectin-1 as a key alarmin at the maternal-fetal interface
- PMID: 37475853
- PMCID: PMC10354452
- DOI: 10.3389/fimmu.2023.1196395
A unique maternal and placental galectin signature upon SARS-CoV-2 infection suggests galectin-1 as a key alarmin at the maternal-fetal interface
Abstract
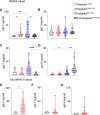
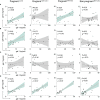
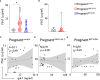
The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pandemic imposed a risk of infection and disease in pregnant women and neonates. Successful pregnancy requires a fine-tuned regulation of the maternal immune system to accommodate the growing fetus and to protect the mother from infection. Galectins, a family of β-galactoside-binding proteins, modulate immune and inflammatory processes and have been recognized as critical factors in reproductive orchestration, including maternal immune adaptation in pregnancy. Pregnancy-specific glycoprotein 1 (PSG1) is a recently identified gal-1 ligand at the maternal-fetal interface, which may facilitate a successful pregnancy. Several studies suggest that galectins are involved in the immune response in SARS-CoV-2-infected patients. However, the galectins and PSG1 signature upon SARS-CoV-2 infection and vaccination during pregnancy remain unclear. In the present study, we examined the maternal circulating levels of galectins (gal-1, gal-3, gal-7, and gal-9) and PSG1 in pregnant women infected with SARS-CoV-2 before vaccination or uninfected women who were vaccinated against SARS-CoV-2 and correlated their expression with different pregnancy parameters. SARS-CoV-2 infection or vaccination during pregnancy provoked an increase in maternal gal-1 circulating levels. On the other hand, levels of PSG1 were only augmented upon SARS-CoV-2 infection. A healthy pregnancy is associated with a positive correlation between gal-1 concentrations and gal-3 or gal-9; however, no correlation was observed between these lectins during SARS-CoV-2 infection. Transcriptome analysis of the placenta showed that gal-1, gal-3, and several PSG and glycoenzymes responsible for the synthesis of gal-1-binding glycotopes (such as linkage-specific N-acetyl-glucosaminyltransferases (MGATs)) are upregulated in pregnant women infected with SARS-CoV-2. Collectively, our findings identify a dynamically regulated "galectin-specific signature" that accompanies the SARS-CoV-2 infection and vaccination in pregnancy, and they highlight a potentially significant role for gal-1 as a key pregnancy protective alarmin during virus infection.
Keywords: COVID-19; PSG1; SARS-CoV-2; galectin-1; galectin-3; galectin-7; galectin-9.
Copyright © 2023 Zhao, Tallarek, Wang, Xie, Diemert, Lu-Culligan, Vijayakumar, Kittmann, Urbschat, Bayo, Arck, Farhadian, Dveksler, Garcia and Blois.
Conflict of interest statement
The authors declare that the research was conducted without any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

