Zebrafish as a model for cardiac disease; Cryo-EM structure of native cardiac thin filaments from Danio Rerio
- PMID: 37480427
- PMCID: PMC10542308
- DOI: 10.1007/s10974-023-09653-5
Zebrafish as a model for cardiac disease; Cryo-EM structure of native cardiac thin filaments from Danio Rerio
Abstract
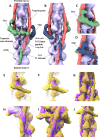
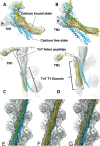
Actin, tropomyosin and troponin, the proteins that comprise the contractile apparatus of the cardiac thin filament, are highly conserved across species. We have used cryo-EM to study the three-dimensional structure of the zebrafish cardiac thin and actin filaments. With 70% of human genes having an obvious zebrafish orthologue, and conservation of 85% of disease-causing genes, zebrafish are a good animal model for the study of human disease. Our structure of the zebrafish thin filament reveals the molecular interactions between the constituent proteins, showing that the fundamental organisation of the complex is the same as that reported in the human reconstituted thin filament. A reconstruction of zebrafish cardiac F-actin demonstrates no deviations from human cardiac actin over an extended length of 14 actin subunits. Modelling zebrafish homology models into our maps enabled us to compare, in detail, the similarity with human models. The structural similarities of troponin-T in particular, a region known to contain a hypertrophic cardiomyopathy 'hotspot', confirm the suitability of zebrafish to study these disease-causing mutations.
Keywords: Actin; Cryo-EM; Thin filament; Tropomyosin; Troponin; Zebrafish.
© 2023. The Author(s).
Conflict of interest statement
I declare that the authors have no competing interests as defined by Springer, or other interests that might be perceived to influence the results and/or discussion reported in this paper.
Figures



Similar articles
-
The role of the troponin T interactions with actin in regulation of cardiac thin filament revealed by the troponin T pathogenic variant Ile79Asn.J Mol Cell Cardiol. 2025 Jul;204:55-67. doi: 10.1016/j.yjmcc.2025.05.005. Epub 2025 May 22. J Mol Cell Cardiol. 2025. PMID: 40412797
-
Cryo-EM structures of cardiac muscle α-actin mutants M305L and A331P give insights into the structural mechanisms of hypertrophic cardiomyopathy.Eur J Cell Biol. 2024 Dec;103(4):151460. doi: 10.1016/j.ejcb.2024.151460. Epub 2024 Oct 1. Eur J Cell Biol. 2024. PMID: 39393252 Free PMC article.
-
C-terminal troponin-I residues trap tropomyosin in the muscle thin filament blocked-state.Biochem Biophys Res Commun. 2021 Apr 30;551:27-32. doi: 10.1016/j.bbrc.2021.03.010. Epub 2021 Mar 11. Biochem Biophys Res Commun. 2021. PMID: 33714756 Free PMC article.
-
Troponin Revealed: Uncovering the Structure of the Thin Filament On-Off Switch in Striated Muscle.Biophys J. 2021 Jan 5;120(1):1-9. doi: 10.1016/j.bpj.2020.11.014. Epub 2020 Nov 20. Biophys J. 2021. PMID: 33221250 Free PMC article. Review.
-
Invited Review: pathophysiology of cardiac muscle contraction and relaxation as a result of alterations in thin filament regulation.J Appl Physiol (1985). 2001 Mar;90(3):1125-36. doi: 10.1152/jappl.2001.90.3.1125. J Appl Physiol (1985). 2001. PMID: 11181629 Review.
Cited by
-
John Squire: a leader and seminal contributor to experimental and theoretical muscle research for over 50 years.J Muscle Res Cell Motil. 2023 Sep;44(3):123-124. doi: 10.1007/s10974-023-09659-z. J Muscle Res Cell Motil. 2023. PMID: 37740044 No abstract available.
References
Publication types
MeSH terms
Substances
Grants and funding
- FS/14/18/3071/BHF_/British Heart Foundation/United Kingdom
- MR/W003309/2/MRC_/Medical Research Council/United Kingdom
- BB/R000484/1/BB_/Biotechnology and Biological Sciences Research Council/United Kingdom
- FS/20/5/34973/BHF_/British Heart Foundation/United Kingdom
- CH/17/1/32804/BHF_/British Heart Foundation/United Kingdom
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

