Endoplasmic reticulum stress controls PIN-LIKES abundance and thereby growth adaptation
- PMID: 37487064
- PMCID: PMC10400986
- DOI: 10.1073/pnas.2218865120
Endoplasmic reticulum stress controls PIN-LIKES abundance and thereby growth adaptation
Abstract
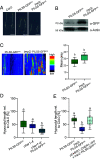
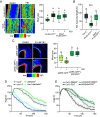
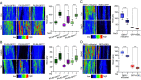
Extreme environmental conditions eventually limit plant growth [J. R. Dinneny, Annu. Rev. Cell Dev. Biol. 35, 1-19 (2019), N. Gigli-Bisceglia, C. Testerink, Curr. Opin. Plant Biol. 64, 102120 (2021)]. Here, we reveal a mechanism that enables multiple external cues to get integrated into auxin-dependent growth programs in Arabidopsis thaliana. Our forward genetics approach on dark-grown hypocotyls uncovered that an imbalance in membrane lipids enhances the protein abundance of PIN-LIKES (PILS) [E. Barbez et al., Nature 485, 119 (2012)] auxin transport facilitators at the endoplasmic reticulum (ER), which thereby limits nuclear auxin signaling and growth rates. We show that this subcellular response relates to ER stress signaling, which directly impacts PILS protein turnover in a tissue-dependent manner. This mechanism allows PILS proteins to integrate environmental input with phytohormone auxin signaling, contributing to stress-induced growth adaptation in plants.
Keywords: PIN-LIKES; auxin; endoplasmic reticulum; membrane lipids; protein turnover.
Conflict of interest statement
The authors declare no competing interest.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

