Improving resource utilisation in SLE drug development through innovative trial design
- PMID: 37491104
- PMCID: PMC10373732
- DOI: 10.1136/lupus-2022-000890
Improving resource utilisation in SLE drug development through innovative trial design
Abstract
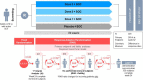
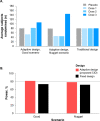
SLE is a complex autoimmune disease with considerable unmet need. Numerous clinical trials designed to investigate novel therapies are actively enrolling patients straining limited resources and creating inefficiencies that increase enrolment challenges. This has motivated investigators developing novel drugs and treatment strategies to consider innovative trial designs that aim to improve the efficiency of generating evidence; these strategies propose conducting fewer trials, involving smaller numbers of patients, while maintaining scientific rigour in safety and efficacy data collection and analysis. In this review we present the design of two innovative phase IIb studies investigating efavaleukin alfa and rozibafusp alfa for the treatment of SLE which use an adaptive study design. This design was selected as a case study, investigating efavaleukin alfa, in the Food and Drug Administration's Complex Innovative Trial Design Pilot Program. The adaptive design approach includes prospectively planned modifications at predefined interim timepoints. Interim assessments of futility allow for a trial to end early when the investigational therapy is unlikely to provide meaningful treatment benefits to patients, which can release eligible patients to participate in other-potentially more promising-trials, or seek alternative treatments. Response-adaptive randomisation allows randomisation ratios to change based on accumulating data, in favour of the more efficacious dose arm(s), while the study is ongoing. Throughout the trial the placebo arm allocation ratio is maintained constant. These design elements can improve the statistical power in the estimation of treatment effect and increase the amount of safety and efficacy data collected for the optimal dose(s). Furthermore, these trials can provide the required evidence to potentially serve as one of two confirmatory trials needed for regulatory approval. This can reduce the need for multiple phase III trials, the total patient requirements, person-exposure risk, and ultimately the time and cost of investigational drug development programmes.
Trial registration: ClinicalTrials.gov NCT04058028 NCT04680637.
Keywords: Adaptive Clinical Trial Design; Autoimmune Diseases; Lupus Erythematosus, Systemic.
© Author(s) (or their employer(s)) 2023. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: SG, EK, MM, CEM are employees and stockholders of Amgen. JTM is a consultant for AbbVie, Alexion, Amgen, AstraZeneca, Aurinia Pharmaceuticals, BMS, EMD Serono, Gilead, Genentech, GSK, Lilly, Merck, Pfizer, Provention Bio, RemeGen, Sanofi, UCB, and Zenas, is a speaker for AbbVie, Biogen, Sanofi, and RemeGen, and has received grants (to institution) from AstraZeneca, BMS, and GSK. ADA is an investigator and consultant for AbbVie, Amgen, AstraZeneca, Aurinia Pharmaceuticals, BMS, Celgene, Idorsia, Genentech, GSK, Janssen, Lilly, Mallinckrodt Pharmaceuticals, Pfizer, and UCB. KK is a consultant for AbbVie, AstraZeneca, Biogen, BMS, Cabaletta Bio, EMD Serono, Equillium, Genentech, Gilead, GSK, Kangpu Biopharmaceuticals, Kezar Life Sciences, Kyowa Kirin Co., and Merck, and has received research support from Acceleron Pharma, Alexion, Alpine Immune Sciences, Amgen, Horizon, Idorsia, Kyowa Kirin Co., Lupus Research Alliance and the Wolfe Family Program in Lupus, NIH, Novartis, Provention Bio, UCB, and Vera Therapeutics.
Figures