Long-Term Survival, Prognostic Factors, and Selection of Patients With Colorectal Cancer for Liver Transplant: A Nonrandomized Controlled Trial
- PMID: 37494056
- PMCID: PMC10372758
- DOI: 10.1001/jamasurg.2023.2932
Long-Term Survival, Prognostic Factors, and Selection of Patients With Colorectal Cancer for Liver Transplant: A Nonrandomized Controlled Trial
Abstract
Importance: Liver transplant for colorectal cancer with liver metastases was abandoned in the 1990s due to poor overall survival. From 2006, liver transplant for in nonresectable colorectal liver metastases has been reexamined through different prospective trials.
Objective: To determine predictive factors for transplant long-term survival and cure after liver transplant.
Design, setting, and participants: This was a prospective, nonrandomized controlled cohort study derived from different clinical trials on liver transplant for colorectal liver metastases from 2006 to 2020 at Oslo University Hospital. The trials differed in prognostic inclusion criteria, but the design was otherwise identical regarding follow-up scheme to determine disease recurrence, overall survival, and survival after relapse. Final data analysis was performed on December 31, 2021. All patients with colorectal liver metastases from comparable prospective liver transplant studies were included.
Exposure: Liver transplant.
Main outcomes and measures: Disease-free survival, overall survival, and survival time after recurrence were determined in all participants.
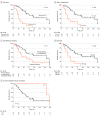
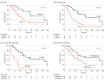
Results: A total of 61 patients (median [range] age, 57.8 [28.7-71.1] years; 35 male [57.4%]) underwent liver transplant at Oslo University Hospital. Posttransplant observation time ranged from 16 to 165 months, and no patient was lost to follow-up. Median disease-free period, overall survival, and survival after relapse were 11.8 (95% CI, 9.3-14.2) months, 60.3 (95% CI, 44.3-76.4) months, and 37.1 (95% CI, 4.6-69.5) months, respectively. Negative predictive factors for overall survival included the following: largest tumor size greater than 5.5 cm (median OS, 25.3 months; 95% CI, 15.8-34.8 months; P <.001), progressive disease while receiving chemotherapy (median OS, 39.8 months; 95% CI, 28.8-50.7 months; P = .02), plasma carcinoembryonic antigen values greater than 80 μg/L (median OS, 26.6 months; 95% CI, 22.7-30.6 months; P <.001), liver metabolic tumor volume on positron emission tomography of greater than 70 cm3 (26.6 months; 95% CI, 11.8-41.5 months; P <.001), primary tumor in the ascending colon (17.9 months; 95% CI, 0-37.5 months; P <.001), tumor burden score of 9 or higher (23.3 months; 95% CI, 19.2-27.4 months; P = .02), and 9 or more liver lesions (42.5 months; 95% CI, 17.2-67.8 months; P = .02). An Oslo score of 0 or Fong Clinical Risk Score of 1 yielded 10-year survival of 88.9% and 80.0%, respectively.
Conclusions and relevance: Results of this nonrandomized controlled trial suggest that selected patients with liver-only metastases and favorable pretransplant prognostic scoring had long-term survival comparable with conventional indications for liver transplant, thus providing a potential curative treatment option in patients otherwise offered only palliative care.
Conflict of interest statement
Figures

Comment in
-
Who Should Undergo Transplant for Unresectable Colorectal Liver Metastases-Finding the Needle in the Haystack.JAMA Surg. 2023 Sep 1;158(9):e232933. doi: 10.1001/jamasurg.2023.2933. Epub 2023 Sep 13. JAMA Surg. 2023. PMID: 37494008 No abstract available.
-
Resection Details, Donor Segment Volume, and Adjuvant Chemotherapy in Patients With Colorectal Cancer and Liver Transplant.JAMA Surg. 2024 Mar 1;159(3):350. doi: 10.1001/jamasurg.2023.5799. JAMA Surg. 2024. PMID: 37966835 No abstract available.
-
Liver transplantation for colorectal liver metastasis: the exception, not the rule.Hepatobiliary Surg Nutr. 2024 Oct 1;13(5):857-860. doi: 10.21037/hbsn-24-367. Epub 2024 Aug 26. Hepatobiliary Surg Nutr. 2024. PMID: 39507732 Free PMC article. No abstract available.
References
-
- Nordlinger B, Sorbye H, Glimelius B, et al. ; EORTC Gastro-Intestinal Tract Cancer Group; Cancer Research UK; Arbeitsgruppe Lebermetastasen und–tumoren in der Chirurgischen Arbeitsgemeinschaft Onkologie (ALM-CAO); Australasian Gastro-Intestinal Trials Group (AGITG); Fédération Francophone de Cancérologie Digestive (FFCD) . Perioperative FOLFOX4 chemotherapy and surgery vs surgery alone for resectable liver metastases from colorectal cancer (EORTC 40983): long-term results of a randomized, controlled, phase 3 trial. Lancet Oncol. 2013;14(12):1208-1215. doi:10.1016/S1470-2045(13)70447-9 - DOI - PubMed
-
- Van Cutsem E, Köhne CH, Láng I, et al. . Cetuximab plus irinotecan, fluorouracil, and leucovorin as first-line treatment for metastatic colorectal cancer: updated analysis of overall survival according to tumor KRAS and BRAF mutation status. J Clin Oncol. 2011;29(15):2011-2019. doi:10.1200/JCO.2010.33.5091 - DOI - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

