Efficacy and Potential Positioning of Tezepelumab in the Treatment of Severe Asthma
- PMID: 37496871
- PMCID: PMC10369521
- DOI: 10.1016/j.opresp.2022.100231
Efficacy and Potential Positioning of Tezepelumab in the Treatment of Severe Asthma
Abstract
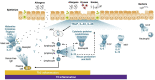
The excellent results for monoclonal antibodies in the treatment of severe uncontrolled asthma (SUCA) represent a milestone in current treatment of asthmatic disorders. Remaining, however, are several subsidiary areas for improvement in which new biologics are expected to make a decisive contribution. These biologics include tezepelumab, a monoclonal antibody that blocks thymic stromal lymphopoietin (TSLP). TSLP is an epithelial-release cytokine (alarmin) that plays a key role in initiating both the innate (group 2 innate lymphoid cell (ILC) pathway) and the acquired (T helper 2 (Th2) pathway) immune responses by activating the type 2 (T2) asthma inflammatory pathway through both. It is also thought that it may additionally intervene in the neutrophilic non-T2 inflammatory pathway (via interaction with ILC3 and interleukin-17). Six clinical trials that included 2187 patients with uncontrolled asthma, with 2 or more exacerbations in the previous year, on medium/high-dose inhaled corticosteroids and at least 1 other controller, have demonstrated - irrespective of T2 endotype (and possibly also non-T2 endotype) - the efficacy and safety of tezepelumab, as it significantly reduces exacerbations (61.7%-66%) and bronchial hyperresponsiveness, and improves lung function, disease control, and quality of life. Tezepelumab could be indicated for the treatment of patients with, independently of the T2 phenotype (eosinophilic and non-eosinophilic), and may even be the only biologic available for treatment of non-T2 SUCA.
Los excelentes resultados de los anticuerpos monoclonales en el tratamiento del asma grave no controlada (AGNC) constituyen un hito en el tratamiento actual de los trastornos asmáticos. Sin embargo, aún quedan varios aspectos complementarios susceptibles de mejorar para los que se esperan contribuciones decisivas de los nuevos biofármacos, entre los cuales se encuentra el tezepelumab, un anticuerpo monoclonal que bloquea la linfopoyetina estromal tímica (TSLP). La TSLP es una citocina de liberación epitelial (alarmina) que desempeña una función clave en el inicio de las respuestas inmunitarias tanto innata (vía de las células linfocíticas innatas [ILC] del grupo 2) como adaptativa (vía de los linfocitos T cooperadores 2 [Th2]), activando la vía inflamatoria del asma del tipo 2 (T2) mediante ambas. También se cree que puede intervenir en la vía inflamatoria neutrofílica con T2 baja (mediante la interacción con los ILC3 y la interleucina 17). En seis ensayos clínicos que incluyeron a 2.187 pacientes con asma no controlada, dos o más exacerbaciones en el año anterior, a tratamiento con corticosteroides inhalados en dosis medias o altas y con un mínimo de un tratamiento preventivo adicional, se ha demostrado la eficacia y seguridad del tezepelumab sin importar el endotipo T2 (y posiblemente tampoco el endotipo no T2), ya que reduce significativamente las exacerbaciones (61,7-66%) y la hiperreactividad bronquial y mejora la función pulmonar, el control de la enfermedad y la calidad de vida. El tezepelumab puede estar indicado para tratar a pacientes con asma grave, independientemente del fenotipo T2 (eosinofílico y no eosinofílico), y tal vez sea incluso el único biofármaco existente para el tratamiento del AGNC no T2.
Keywords: Alarmin; Phenotype; Thymic stromal lymphopoietin; Uncontrolled asthma.
© 2023 Sociedad Española de Neumología y Cirugía Torácica (SEPAR). Published by Elsevier España, S.L.U.
Figures
Similar articles
-
Tezepelumab for Severe Asthma: One Drug Targeting Multiple Disease Pathways and Patient Types.J Asthma Allergy. 2024 Mar 19;17:219-236. doi: 10.2147/JAA.S342391. eCollection 2024. J Asthma Allergy. 2024. PMID: 38524099 Free PMC article. Review.
-
Unmet need in severe, uncontrolled asthma: can anti-TSLP therapy with tezepelumab provide a valuable new treatment option?Respir Res. 2020 Oct 15;21(1):268. doi: 10.1186/s12931-020-01505-x. Respir Res. 2020. PMID: 33059715 Free PMC article. Review.
-
Tezepelumab: a novel biological therapy for the treatment of severe uncontrolled asthma.Expert Opin Investig Drugs. 2019 Nov;28(11):931-940. doi: 10.1080/13543784.2019.1672657. Epub 2019 Oct 10. Expert Opin Investig Drugs. 2019. PMID: 31549891 Review.
-
NAVIGATOR: a phase 3 multicentre, randomized, double-blind, placebo-controlled, parallel-group trial to evaluate the efficacy and safety of tezepelumab in adults and adolescents with severe, uncontrolled asthma.Respir Res. 2020 Oct 13;21(1):266. doi: 10.1186/s12931-020-01526-6. Respir Res. 2020. PMID: 33050934 Free PMC article. Clinical Trial.
-
CASCADE: a phase 2, randomized, double-blind, placebo-controlled, parallel-group trial to evaluate the effect of tezepelumab on airway inflammation in patients with uncontrolled asthma.Respir Res. 2020 Oct 13;21(1):265. doi: 10.1186/s12931-020-01513-x. Respir Res. 2020. PMID: 33050900 Free PMC article.
Cited by
-
Eosinophil-Driven vs. Eosinophil-Associated Severe Asthma: Practical Implications for Target Treatment.Int J Mol Sci. 2025 Feb 18;26(4):1729. doi: 10.3390/ijms26041729. Int J Mol Sci. 2025. PMID: 40004192 Free PMC article. Review.
-
Vitamin D3 suppresses NLRP3 inflammasome pathway and enhances steroid sensitivity in a neutrophilic steroid hyporesponsive asthma mouse model.Inflamm Res. 2025 Mar 14;74(1):51. doi: 10.1007/s00011-025-02009-4. Inflamm Res. 2025. PMID: 40082319
-
Personalized and Precision Medicine in Asthma and Eosinophilic Esophagitis: The Role of T2 Target Therapy.Pharmaceutics. 2023 Sep 21;15(9):2359. doi: 10.3390/pharmaceutics15092359. Pharmaceutics. 2023. PMID: 37765327 Free PMC article. Review.
-
Clinical and Pathophysiological Tangles Between Allergy and Autoimmunity: Deconstructing an Old Dichotomic Paradigm.Clin Rev Allergy Immunol. 2025 Feb 11;68(1):13. doi: 10.1007/s12016-024-09020-3. Clin Rev Allergy Immunol. 2025. PMID: 39932658 Free PMC article. Review.
-
Positioning tezepelumab for patients with severe asthma: from evidence to unmet needs.J Int Med Res. 2024 Nov;52(11):3000605241297532. doi: 10.1177/03000605241297532. J Int Med Res. 2024. PMID: 39552062 Free PMC article. Review.
References
-
- Global Initiative for Asthma (GINA) 2022. Global strategy for asthma management and prevention. Available from: www.ginasthma.org.
-
- 2022. GEMA 5.2. Guía Española para el Manejo del Asma. Available from: http://www.gemasma.com [accessed May 2022] - PubMed
-
- Quirce S., Plaza V., Picado C., Vennera M., Casafont J. Prevalence of uncontrolled severe persistent asthma in pneumology and allergy hospital units in Spain. J Invest Allergol Clin Immunol. 2011;21:466–471. [PMID: 21995180] - PubMed
-
- Plaza-Martín A.M., Vennera M.C., Galera J., Herráez L., PREX Study Group Prevalence and clinical profile of difficult-to-control severe asthma in children: results from pneumology and allergy hospital units in Spain. Allergol Immunopathol (Madr) 2014;42:510–517. doi: 10.1016/j.aller.2014.02.003. [Epub 16.06.14; PMID: 24948187] - DOI - PubMed
-
- Martínez-Moragón E., Serra-Batllés J., De Diego A., Palop M., Casan P., Rubio-Terrés C., et al. Coste económico del paciente asmático en España (estudio AsmaCost) Economic cost of treating the patient with asthma in Spain: the AsmaCost studyArch Bronconeumol. 2009;45:481–486. doi: 10.1016/j.arbres.2009.04.006. [in Spanish; Epub 13.06.09; PMID: 19525051] - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources

