Inflammatory macrophages exploited by oral streptococcus increase IL-1B release via NLRP6 inflammasome
- PMID: 37497744
- PMCID: PMC10533225
- DOI: 10.1093/jleuko/qiad089
Inflammatory macrophages exploited by oral streptococcus increase IL-1B release via NLRP6 inflammasome
Abstract
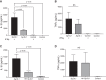
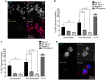
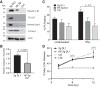
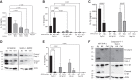
Chronic inflammatory periodontal disease develops in part from the infiltration of a large number of classically activated inflammatory macrophages that release inflammatory cytokines important for disease progression, including inflammasome-dependent interleukin (IL)-1β. Streptococcus gordonii is a normally commensal oral microorganism; while not causative, recent evidence indicates that commensal oral microbes are required for the full development of periodontal disease. We have recently reported that inflammatory macrophages counterintuitively allow for the increased survival of phagocytosed S. gordonii over nonactivated or alternatively activated macrophages. This survival is dependent on increased reactive oxygen species production within the phagosome of the inflammatory macrophages, and resistance by the bacterium and can result in S. gordonii damaging the phagolysosomes. Here, we show that activated macrophages infected with live S. gordonii release more IL-1β than non-activated macrophages infected with either live or dead S. gordonii, and that the survival of oral Streptococci are more dependent on macrophage activation than other Gram positive microbes, both classical pathogens and commensals. We also find that S. gordonii-dependent inflammatory macrophage inflammasome activation requires the cytoplasmic NLRP6. Overall, our results suggest S. gordonii is capable of evading immune destruction, increasing inflammatory mediators, and increasing inflammatory macrophage response, and that this ability is increased under conditions of inflammation. This work reveals additional mechanisms by which normally commensal oral streptococci-macrophage interactions can change, resulting in increased release of mature IL-1β, potentially contributing to an environment that perpetuates inflammation.
Keywords: commensal; inflammasome; macrophages; streptococcus gordonii.
© The Author(s) 2023. Published by Oxford University Press on behalf of Society for Leukocyte Biology. All rights reserved. For permissions, please e-mail: journals.permissions@oup.com.
Conflict of interest statement
Conflict of interest statement. The authors declare no conflict of interest.
Figures

Similar articles
-
Macrophage Polarization Alters Postphagocytosis Survivability of the Commensal Streptococcus gordonii.Infect Immun. 2018 Feb 20;86(3):e00858-17. doi: 10.1128/IAI.00858-17. Print 2018 Mar. Infect Immun. 2018. PMID: 29229734 Free PMC article.
-
Spent culture supernatant of Streptococcus gordonii mitigates inflammation of human periodontal cells and inhibits proliferation of pathogenic oral microbes.J Periodontol. 2023 Apr;94(4):575-585. doi: 10.1002/JPER.22-0333. Epub 2023 Jan 6. J Periodontol. 2023. PMID: 36369979 Free PMC article.
-
The oral commensal, Streptococcus gordonii, synergizes with Tat protein to induce HIV-1 promoter activation in monocytes/macrophages.Cell Immunol. 2011;269(1):38-45. doi: 10.1016/j.cellimm.2011.03.008. Epub 2011 Mar 15. Cell Immunol. 2011. PMID: 21459369 Free PMC article.
-
Caspase-11 non-canonical inflammasome: a critical sensor of intracellular lipopolysaccharide in macrophage-mediated inflammatory responses.Immunology. 2017 Oct;152(2):207-217. doi: 10.1111/imm.12787. Epub 2017 Jul 31. Immunology. 2017. PMID: 28695629 Free PMC article. Review.
-
Role of interleukin-1 and inflammasomes in oral disease.J Oral Biosci. 2020 Sep;62(3):242-248. doi: 10.1016/j.job.2020.07.003. Epub 2020 Aug 6. J Oral Biosci. 2020. PMID: 32771408 Review.
Cited by
-
NLRP3 inflammasome activity and periodontal disease pathogenesis-A bidirectional relationship.Oral Dis. 2024 Oct;30(7):4069-4077. doi: 10.1111/odi.15005. Epub 2024 May 30. Oral Dis. 2024. PMID: 38817019 Free PMC article. Review.
-
The oral microbiome and oral and upper gastrointestinal diseases.J Oral Microbiol. 2024 Jun 3;16(1):2355823. doi: 10.1080/20002297.2024.2355823. eCollection 2024. J Oral Microbiol. 2024. PMID: 38835339 Free PMC article. Review.
References
-
- Gonzalez OA, Novak MJ, Kirakodu S, Stromberg A, Nagarajan R, Huang CB, Chen KC, Orraca L, Martinez-Gonzalez J, Ebersole JL. Differential gene expression profiles reflecting macrophage polarization in aging and periodontitis gingival tissues. Immunol Invest. 2015:44(7):643–664. 10.3109/08820139.2015.1070269 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

