Adverse effects of gestational ω-3 and ω-6 polyunsaturated fatty acid imbalance on the programming of fetal brain development
- PMID: 37497857
- PMCID: PMC10909496
- DOI: 10.1111/jne.13320
Adverse effects of gestational ω-3 and ω-6 polyunsaturated fatty acid imbalance on the programming of fetal brain development
Abstract
Obesity is a key medical challenge of our time. The increasing number of children born to overweight or obese women is alarming. During pregnancy, the circulation of the mother and her fetus interact to maintain the uninterrupted availability of essential nutrients for fetal organ development. In doing so, the mother's dietary preference determines the amount and composition of nutrients reaching the fetus. In particular, the availability of polyunsaturated fatty acids (PUFAs), chiefly their ω-3 and ω-6 subclasses, can change when pregnant women choose a specific diet. Here, we provide a succinct overview of PUFA biochemistry, including exchange routes between ω-3 and ω-6 PUFAs, the phenotypes, and probable neurodevelopmental disease associations of offspring born to mothers consuming specific PUFAs, and their mechanistic study in experimental models to typify signaling pathways, transcriptional, and epigenetic mechanisms by which PUFAs can imprint long-lasting modifications to brain structure and function. We emphasize that the ratio, rather than the amount of individual ω-3 or ω-6 PUFAs, might underpin physiologically correct cellular differentiation programs, be these for neurons or glia, during pregnancy. Thereupon, the PUFA-driven programming of the brain is contextualized for childhood obesity, metabolic, and endocrine illnesses.
Keywords: arachidonic acid; dietary requirement; neurodevelopmental disorder; neuronal wiring; nutrient conversion.
© 2023 The Authors. Journal of Neuroendocrinology published by John Wiley & Sons Ltd on behalf of British Society for Neuroendocrinology.
Conflict of interest statement
The authors of this manuscript declare no conflict of interest.
Figures
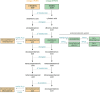

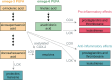
References
-
- Barker DJ. In utero programming of chronic disease. Clin Sci. 1998;95(2):115‐128. - PubMed
-
- Faa G, Manchia M, Pintus R, Gerosa C, Marcialis MA, Fanos V. Fetal programming of neuropsychiatric disorders. Birth Defects Res C Embryo Today. 2016;108(3):207‐223. - PubMed
-
- Rubino T, Parolaro D. The impact of exposure to cannabinoids in adolescence: insights from animal models. Biol Psychiatry. 2016;79(7):578‐585. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

