Treatment of crow's feet lines and forehead lines with Botox (onabotulinumtoxinA): Development, insights, and impact
- PMID: 37499083
- PMCID: PMC10374187
- DOI: 10.1097/MD.0000000000032496
Treatment of crow's feet lines and forehead lines with Botox (onabotulinumtoxinA): Development, insights, and impact
Abstract
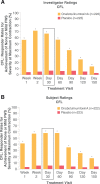
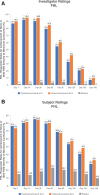
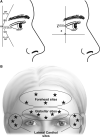
Extrinsic and age-related intrinsic factors contribute to the development of facial lines, including lateral canthal lines (called crow's feet lines [CFL]) and horizontal forehead lines (FHL). OnabotulinumtoxinA is a highly effective treatment for facial lines that inhibits acetylcholine release at the neuromuscular junction. This temporary chemical denervation leads to localized muscle relaxation and subsequent wrinkle reduction. Early studies of onabotulinumtoxinA treatment for facial neuronal disorders such as dystonia documented improvements in FHL and CFL. After the neurotoxin was approved for treating frown lines (glabellar lines [GL]), individuals requested treatment for other rhytids, and physicians continued assessing use in new areas. Once onabotulinumtoxinA was in clinical trial development, its efficacy and safety for CFL and FHL were successively evaluated as required by the US Food and Drug Administration and by key global health authorities, including those in the European Union, Japan, and China. Allergan, collaborating with leading physicians, established clinical programs that included novel safety and efficacy measures to meet regulatory requirements. Global, phase 3, randomized, controlled studies of CFL and FHL met rigorous primary endpoints. Some countries mandated clinical trial data beyond US and European regulations, and Allergan conducted 11 studies in total, fulfilling diverse regulatory and study population data requirements. Adverse events associated with local spread, including brow and eyelid ptosis, diplopia, headache, and eyelid sensory disorder, were infrequent and well tolerated. Consequently, onabotulinumtoxinA treatment of upper facial lines is now established globally as a highly effective, minimally invasive treatment for patients to achieve a natural appearance and look younger.
Copyright © 2023 the Author(s). Published by Wolters Kluwer Health, Inc.
Figures
References
-
- Small R. Botulinum toxin injection for facial wrinkles. Am Fam Physician. 2014;90:168–75. - PubMed
-
- Carruthers A, Bruce S, Cox SE, et al. . OnabotulinumtoxinA for treatment of moderate to severe crow’s feet lines: a review. Aesthet Surg J. 2016;36:591–7. - PubMed
-
- Beer K, Beer J. Overview of facial aging. Facial Plast Surg. 2009;25:281–4. - PubMed
-
- Sundaram H, Signorini M, Liew S, et al. . Global Aesthetics Consensus Group. Global Aesthetics Consensus: botulinum toxin type 1—evidence-based review, emerging concepts, and consensus recommendations for aesthetic use, including updates on complications. Plast Reconstr Surg. 2016;137:518e–29e. - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

