On-Demand Free Radical Release by Laser Irradiation for Photothermal-Thermodynamic Biofilm Inactivation and Tooth Whitening
- PMID: 37504433
- PMCID: PMC10379348
- DOI: 10.3390/gels9070554
On-Demand Free Radical Release by Laser Irradiation for Photothermal-Thermodynamic Biofilm Inactivation and Tooth Whitening
Abstract
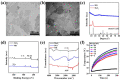
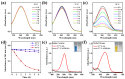
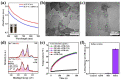
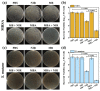
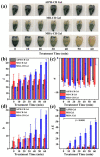
Dental diseases associated with biofilm infections and tooth staining affect billions of people worldwide. In this study, we combine photothermal agents (MoS2@BSA nanosheets, MB NSs), a thermolysis free-radical initiator (AIPH), and carbomer gel to develop laser-responsive hydrogel (MBA-CB Gel) for biofilm inactivating and tooth whitening. Under a physiological temperature without laser irradiation, MB NSs can eliminate free radicals generated from the slow decomposition of AIPH due to their antioxidative activity, thereby avoiding potential side effects. A cytotoxicity study indicates that MB NSs can protect mammalian cells from the free radicals released from AIPH without laser irradiation. Upon exposure to laser irradiation, MB NSs promote the rapid decomposition of AIPH to release free radicals by photothermal effect, suggesting their on-demand release ability of free radicals. In vitro experimental results show that the bacteria inactivation efficiency is 99.91% (3.01 log units) for planktonic Streptococcus mutans (S. mutans) and 99.98% (3.83 log units) for planktonic methicillin-resistant Staphylococcus aureus (MRSA) by the mixed solution of MB NSs and AIPH (MBA solution) under 808 nm laser irradiation (1.0 W/cm2, 5 min). For S. mutans biofilms, an MBA solution can inactivate 99.97% (3.63 log units) of the bacteria under similar laser irradiation conditions. Moreover, MBA-CB Gel can whiten an indigo carmine-stained tooth under laser irradiation after 60 min of laser treatment, and the color difference (ΔE) in the teeth of the MBA-CB Gel treatment group was 10.9 times that of the control group. This study demonstrates the potential of MBA-CB Gel as a promising platform for biofilm inactivation and tooth whitening. It is worth noting that, since this study only used stained models of extracted teeth, the research results may not fully reflect the actual clinic situation. Future clinical research needs to further validate these findings.
Keywords: bacterial biofilm; controllable free radical release; photothermal-thermodynamic therapy; tooth whitening; transition-metal dichalcogenides.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Thermo-responsive and biodegradable MoS2-based nanoplatform for tumor therapy and postoperative wound management.J Colloid Interface Sci. 2025 May 15;686:634-649. doi: 10.1016/j.jcis.2025.01.257. Epub 2025 Jan 31. J Colloid Interface Sci. 2025. PMID: 39914308
-
NIR-responsive MoS2-Cu2WS4 nanosheets for catalytic/photothermal therapy of methicillin-resistant Staphylococcus aureus infections.Nanoscale. 2022 Jul 14;14(27):9796-9805. doi: 10.1039/d2nr01597h. Nanoscale. 2022. PMID: 35770918
-
Injectable Hydrogel Containing TiO Nanosheets for Synergistic Photothermal/Thermodynamic Therapy.ACS Appl Mater Interfaces. 2023 Jul 26;15(29):34436-34450. doi: 10.1021/acsami.3c04354. Epub 2023 Jul 7. ACS Appl Mater Interfaces. 2023. PMID: 37415554
-
Treatment of triple negative breast cancer by near infrared light triggered mild-temperature photothermal therapy combined with oxygen-independent cytotoxic free radicals.Acta Biomater. 2022 Aug;148:218-229. doi: 10.1016/j.actbio.2022.06.011. Epub 2022 Jun 12. Acta Biomater. 2022. PMID: 35705171
-
Bifunctional defect mediated direct Z-scheme g-C3N4-x/Bi2O3-y heterostructures with enhanced piezo-photocatalytic properties for efficient tooth whitening and biofilm eradication.J Mater Chem B. 2023 Aug 2;11(30):7103-7116. doi: 10.1039/d3tb01044a. J Mater Chem B. 2023. PMID: 37417809
Cited by
-
Internal bleaching with calcium oxalate and laser irradiation for managing discolorations induced by mineral trioxide aggregate.BMC Oral Health. 2025 Mar 7;25(1):349. doi: 10.1186/s12903-025-05703-1. BMC Oral Health. 2025. PMID: 40055702 Free PMC article.
References
-
- GBD 2017 Oral Disorders Collaborators. Bernabe E., Marcenes W., Hernandez C.R., Bailey J., Abreu L.G., Alipour V., Amini S., Arabloo J., Arefi Z., et al. Global, Regional, and National Levels and Trends in Burden of Oral Conditions from 1990 to 2017: A Systematic Analysis for the Global Burden of Disease 2017 Study. J. Dent. Res. 2020;99:362–373. doi: 10.1177/0022034520908533. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

