Effect of Arsenic Soil Contamination on Stress Response Metabolites, 5-Methylcytosine Level and CDC25 Expression in Spinach
- PMID: 37505533
- PMCID: PMC10383220
- DOI: 10.3390/toxics11070568
Effect of Arsenic Soil Contamination on Stress Response Metabolites, 5-Methylcytosine Level and CDC25 Expression in Spinach
Abstract
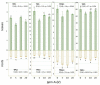
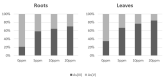
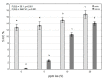
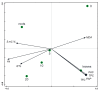
Experimental spinach plants grown in soil with (5, 10 and 20 ppm) arsenic (As) contamination were sampled in 21 days after As(V) contamination. Levels of As in spinach samples (from 0.31 ± 0.06 µg g-1 to 302.69 ± 11.83 µg g-1) were higher in roots and lower in leaves, which indicates a low ability of spinach to translocate As into leaves. Species of arsenic, As(III) and As(V), were represented in favor of the As (III) specie in contaminated variants, suggesting enzymatic arsenate reduction. In relation to predominant As accumulation in roots, changes in malondialdehyde levels were observed mainly in roots, where they decreased significantly with growing As contamination (from 11.97 ± 0.54 µg g-1 in control to 2.35 ± 0.43 µg g-1 in 20 ppm As). Higher values in roots than in leaves were observed in the case of 5-methylcytosine (5-mC). Despite that, a change in 5-mC by As contamination was further deepened in leaves (from 0.20 to 14.10%). In roots of spinach, expression of the CDC25 gene increased by the highest As contamination compared to the control. In the case of total phenolic content, total flavonoid content, total phenolic acids content and total antioxidant capacity were higher levels in leaves in all values, unlike the roots.
Keywords: CDC25; arsenic species; epigenetics; membrane damage; secondary metabolites; spinach.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- Bradl H.B. Heavy Metals in the Environment: Origin, Interaction and Remediation. Elsevier; Amsterdam, The Netherlands: 2005.
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

