Clodronate Liposome-Mediated Phagocytic Hemocyte Depletion Affects the Regeneration of the Cephalic Tentacle of the Invasive Snail, Pomacea canaliculata
- PMID: 37508422
- PMCID: PMC10376890
- DOI: 10.3390/biology12070992
Clodronate Liposome-Mediated Phagocytic Hemocyte Depletion Affects the Regeneration of the Cephalic Tentacle of the Invasive Snail, Pomacea canaliculata
Abstract
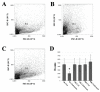
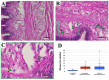
After amputation, granular hemocytes infiltrate the blastema of regenerating cephalic tentacles of the freshwater snail Pomacea canaliculata. Here, the circulating phagocytic hemocytes were chemically depleted by injecting the snails with clodronate liposomes, and the effects on the cephalic tentacle regeneration onset and on Pc-Hemocyanin, Pc-transglutaminase (Pc-TG) and Pc-Allograft Inflammatory Factor-1 (Pc-AIF-1) gene expressions were investigated. Flow cytometry analysis demonstrated that clodronate liposomes targeted large circulating hemocytes, resulting in a transient decrease in their number. Corresponding with the phagocyte depletion, tentacle regeneration onset was halted, and it resumed at the expected pace when clodronate liposome effects were no longer visible. In addition to the regeneration progress, the expressions of Pc-Hemocyanin, Pc-TG, and Pc-AIF-1, which are markers of hemocyte-mediated functions like oxygen transport and immunity, clotting, and inflammation, were modified. After the injection of clodronate liposomes, a specific computer-assisted image analysis protocol still evidenced the presence of granular hemocytes in the tentacle blastema. This is consistent with reports indicating the large and agranular hemocyte population as the most represented among the professional phagocytes of P. canaliculata and with the hypothesis that different hemocyte morphologies could exert diverse biological functions, as it has been observed in other invertebrates.
Keywords: allograft inflammatory factor-1; apple snail; hemocyanin; image analysis; immunity; inflammation; invertebrate; mollusc; phagocytosis; transglutaminase.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of this study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures


References
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

