Yersiniabactin-Producing E. coli Induces the Pyroptosis of Intestinal Epithelial Cells via the NLRP3 Pathway and Promotes Gut Inflammation
- PMID: 37511208
- PMCID: PMC10380849
- DOI: 10.3390/ijms241411451
Yersiniabactin-Producing E. coli Induces the Pyroptosis of Intestinal Epithelial Cells via the NLRP3 Pathway and Promotes Gut Inflammation
Abstract
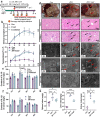
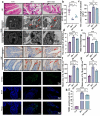
The high-pathogenicity island (HPI) was initially identified in Yersinia and can be horizontally transferred to Escherichia coli to produce yersiniabactin (Ybt), which enhances the pathogenicity of E. coli by competing with the host for Fe3+. Pyroptosis is gasdermin-induced necrotic cell death. It involves the permeabilization of the cell membrane and is accompanied by an inflammatory response. It is still unclear whether Ybt HPI can cause intestinal epithelial cells to undergo pyroptosis and contribute to gut inflammation during E. coli infection. In this study, we infected intestinal epithelial cells of mice with E. coli ZB-1 and the Ybt-deficient strain ZB-1Δirp2. Our findings demonstrate that Ybt-producing E. coli is more toxic and exacerbates gut inflammation during systemic infection. Mechanistically, our results suggest the involvement of the NLRP3/caspase-1/GSDMD pathway in E. coli infection. Ybt promotes the assembly and activation of the NLRP3 inflammasome, leading to GSDMD cleavage into GSDMD-N and promoting the pyroptosis of intestinal epithelial cells, ultimately aggravating gut inflammation. Notably, NLRP3 knockdown alleviated these phenomena, and the binding of free Ybt to NLRP3 may be the trigger. Overall, our results show that Ybt HPI enhances the pathogenicity of E. coli and induces pyroptosis via the NLRP3 pathway, which is a new mechanism through which E. coli promotes gut inflammation. Furthermore, we screened drugs targeting NLRP3 from an existing drug library, providing a list of potential drug candidates for the treatment of gut injury caused by E. coli.
Keywords: E. coli; NLRP3; Ybt HPI; gut inflammation; pyroptosis.
Conflict of interest statement
The authors declare no conflict of interest.
Figures







Similar articles
-
Acinar cell NLRP3 inflammasome and gasdermin D (GSDMD) activation mediates pyroptosis and systemic inflammation in acute pancreatitis.Br J Pharmacol. 2021 Sep;178(17):3533-3552. doi: 10.1111/bph.15499. Epub 2021 May 21. Br J Pharmacol. 2021. PMID: 33871879
-
Induction of macrophage pyroptosis-related factors by pathogenic E. coli high pathogenicity island (HPI) in Yunnan Saba pigs.BMC Vet Res. 2021 Mar 7;17(1):114. doi: 10.1186/s12917-021-02824-x. BMC Vet Res. 2021. PMID: 33678162 Free PMC article.
-
Hemolysin Co-Regulatory Protein 1 Enhances the Virulence of Clinically Isolated Escherichia coli in KM Mice by Increasing Inflammation and Inducing Pyroptosis.Toxins (Basel). 2023 Feb 22;15(3):171. doi: 10.3390/toxins15030171. Toxins (Basel). 2023. PMID: 36977062 Free PMC article.
-
NLRP3 and pyroptosis blockers for treating inflammatory diseases.Trends Pharmacol Sci. 2022 Aug;43(8):653-668. doi: 10.1016/j.tips.2022.04.003. Epub 2022 May 3. Trends Pharmacol Sci. 2022. PMID: 35513901 Review.
-
The mechanisms of NLRP3 inflammasome/pyroptosis activation and their role in Parkinson's disease.Int Immunopharmacol. 2019 Feb;67:458-464. doi: 10.1016/j.intimp.2018.12.019. Epub 2018 Dec 27. Int Immunopharmacol. 2019. PMID: 30594776 Review.
Cited by
-
Yersiniabactin produced by Escherichia coli promotes intestinal inflammation through lipid peroxidation and ferroptosis.Front Microbiol. 2025 Feb 17;16:1542801. doi: 10.3389/fmicb.2025.1542801. eCollection 2025. Front Microbiol. 2025. PMID: 40034497 Free PMC article.
-
Interplay of m6A RNA methylation and gut microbiota in modulating gut injury.Gut Microbes. 2025 Dec;17(1):2467213. doi: 10.1080/19490976.2025.2467213. Epub 2025 Feb 17. Gut Microbes. 2025. PMID: 39960310 Free PMC article. Review.
-
Intestinal luminal anion transporters and their interplay with gut microbiome and inflammation.Am J Physiol Cell Physiol. 2025 May 1;328(5):C1455-C1472. doi: 10.1152/ajpcell.00026.2025. Epub 2025 Mar 6. Am J Physiol Cell Physiol. 2025. PMID: 40047092 Free PMC article. Review.
-
Intestinal mucosal turnover in germ-free piglets infected with E. coli.J Mol Histol. 2024 Dec 4;56(1):24. doi: 10.1007/s10735-024-10278-2. J Mol Histol. 2024. PMID: 39627566 Free PMC article.
-
Multidrug-resistant Escherichia coli causing canine pyometra and urinary tract infections are genetically related but distinct from those causing prostatic abscesses.Sci Rep. 2024 May 24;14(1):11848. doi: 10.1038/s41598-024-62028-9. Sci Rep. 2024. PMID: 38782931 Free PMC article.
References
-
- Ellermann M., Gharaibeh R., Fulbright L., Dogan B., Moore L., Broberg C., Lopez L., Rothemich A., Herzog J., Rogala A., et al. Yersiniabactin-Producing Adherent/Invasive Escherichia coli Promotes Inflammation-Associated Fibrosis in Gnotobiotic Il10-/- Mice. Infect. Immun. 2019;87:e00587-19. doi: 10.1128/IAI.00587-19. - DOI - PMC - PubMed
-
- Dobrindt U., Blum-Oehler G., Nagy G., Schneider G., Johann A., Gottschalk G., Hacker J. Genetic structure and distribution of four pathogenicity islands (PAI I536 to PAI IV536) of uropathogenic Escherichia coli strain 536. Infect. Immun. 2002;70:6365–6372. doi: 10.1128/IAI.70.11.6365-6372.2002. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

