Metabolomics-Based Investigation on the Metabolic Changes in Crassostrea gigas Experimentally Exposed to Galvanic Anodes
- PMID: 37512576
- PMCID: PMC10384061
- DOI: 10.3390/metabo13070869
Metabolomics-Based Investigation on the Metabolic Changes in Crassostrea gigas Experimentally Exposed to Galvanic Anodes
Abstract
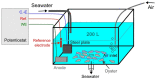
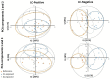
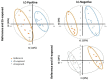
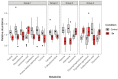
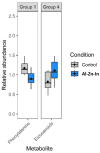
Cathodic protection is widely used to protect metal structures from corrosion in marine environments using sacrificial galvanic anodes. These anodes, either in Zinc, or preferentially nowadays in Al-Zn-In alloys, are expected to corrode instead of the metal structures. This leads to the release of dissolved species, Zn2+, Al3+, and In3+, and solid phases such as Al(OH)3. Few studies have been conducted on their effects on marine organisms, and they concluded that further investigations are needed. We therefore evaluated the effects of Zn and Al-Zn-In anodes on oysters stabulated in tanks, under controlled conditions defined through a comparison with those prevailing in a given commercial seaport used as reference. We analyzed the entire metabolome of gills with a non-targeted metabolomic approach HRMS. A modelling study of the chemical species, corresponding to the degradation products of the anodes, likely to be present near the exposed oysters, was also included. We identified 16 and two metabolites modulated by Zn- and Al-Zn-In-anodes, respectively, that were involved in energy metabolism, osmoregulation, oxidative stress, lipid, nucleotide nucleoside and amino acid metabolisms, defense and signaling pathways. The combination of chemical modelling and metabolomic approach, used here for the first time, enlightened the influence of Zn present in the Al-Zn-In anodes.
Keywords: aluminum; bioaccumulation; biological effects; corrosion products modelling; metabolomics; oysters; sacrificial galvanic anode; zinc.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures
References
-
- Davy H. On the corrosion of copper sheeting by sea water, and on methods of preventing this effect; and on their application to ships of war and other ships. Philos. Trans. R. Soc. Lond. 1824;114:151–158. doi: 10.1080/14786442408644548. - DOI
-
- Lemieux E., Hartt W.H., Lucas K.E. A critical review of Al anode activation and dissolution mechanisms and performance; Proceedings of the Paper NACE-01509, CORROSION 2001 Conference; Houston, TX, USA. 11–16 March 2001.
-
- Muñoz A.G., Saidman S.B., Bessone J.B. Corrosion of an Al–Zn–In alloy in chloride media. Corros. Sci. 2002;44:2171–2182. doi: 10.1016/S0010-938X(02)00042-2. - DOI
-
- Ma J., Wen J., Zhai W., Li Q. In situ corrosion of Al-Zn-In-Mg-Ti-Ce sacrificial anode alloy. Mater. Charac. 2012;65:86–92. doi: 10.1016/j.matchar.2012.01.003. - DOI
-
- Caplat C., Oral R., Mahaut M.-L., Mao A., Barillier D., Guida M., Della Rocca C., Pagano G. Comparative toxicities of aluminum and zinc from sacrificial anodes or from sulfate salt in sea urchin embryos and sperm. Ecotoxicol. Environ. Saf. 2010;73:1138–1143. doi: 10.1016/j.ecoenv.2010.06.024. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

