Flavonoids: Overview of Biosynthesis, Biological Activity, and Current Extraction Techniques
- PMID: 37514347
- PMCID: PMC10384615
- DOI: 10.3390/plants12142732
Flavonoids: Overview of Biosynthesis, Biological Activity, and Current Extraction Techniques
Abstract
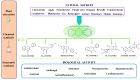
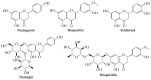
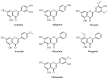
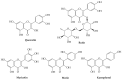
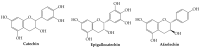
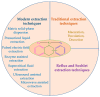
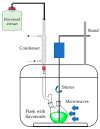
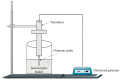
Recently, increased attention has been paid to natural sources as raw materials for the development of new added-value products. Flavonoids are a large family of polyphenols which include several classes based on their basic structure: flavanones, flavones, isoflavones, flavonols, flavanols, and anthocyanins. They have a multitude of biological properties, such as anti-inflammatory, antioxidant, antiviral, antimicrobial, anticancer, cardioprotective, and neuroprotective effects. Current trends of research and development on flavonoids relate to identification, extraction, isolation, physico-chemical characterization, and their applications to health benefits. This review presents an up-to-date survey of the most recent developments in the natural flavonoid classes, the biological activity of representative flavonoids, current extraction techniques, and perspectives.
Keywords: bioactive compounds; biological activity; current extraction techniques; flavonoids; natural compound.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




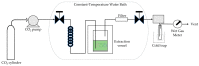


References
-
- Rehan M. Biosynthesis of Diverse Class Flavonoids via Shikimate and Phenylpropanoid Pathway. In: Zepka L.Q., Nascimento T.C.D., Jacob-Lopes E., editors. Bioactive Compounds. IntechOpen; Rijeka, Croatia: 2021. p. Ch. 6.
-
- Pietta P., Minoggio M., Bramati L. Plant Polyphenols: Structure, Occurrence and Bioactivity. In: Rahman A.-u., editor. Studies in Natural Products Chemistry. Volume 28. Elsevier; Amsterdam, The Netherlands: 2003. pp. 257–312.
Publication types
LinkOut - more resources
Full Text Sources

