Development of conserved multi-epitopes based hybrid vaccine against SARS-CoV-2 variants: an immunoinformatic approach
- PMID: 37519944
- PMCID: PMC10374517
- DOI: 10.1007/s40203-023-00156-2
Development of conserved multi-epitopes based hybrid vaccine against SARS-CoV-2 variants: an immunoinformatic approach
Abstract
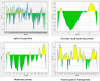
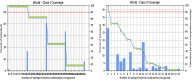
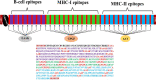
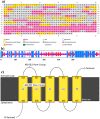
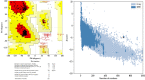
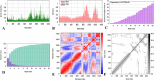
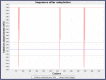
The world has faced unprecedented disruptions like global quarantine and the COVID-19 pandemic due to SARS-CoV-2. To combat these unsettling situations, several effective vaccines have been developed and are currently being used. However, the emergence of new variants due to the high mutation rate of SARS-CoV-2 challenges the efficacy of existing vaccines and has highlighted the need for novel vaccines that will be effective against various SARS-CoV-2 variants. In this study, we exploited the four structural proteins of SARS-CoV-2 to execute a potential multi-epitope vaccine against SARS-CoV-2 and its variants. The vaccine was designed by utilizing the antigenic, non-toxic, and non-allergenic B-cell and T-cell epitopes, which were selected from conserved regions of viral proteins. To build a vaccine construct, epitopes were connected through different linkers and an adjuvant was also attached at the start of the construct to enhance the immunogenicity and specificity of the epitopes. The vaccine construct was then screened through the aforementioned filters and it scored 0.6019 against the threshold of 0.4 on VexiJen 2.0 which validates its antigenicity. Toll-like receptors (i.e., TLR2, TLR3, TLR4, TLR5, and TLR8) and vaccine construct were docked by Cluspro 2.0, and TLR8 showed strong interaction with construct having a maximum negative binding energy of - 1577.1 kCal/mole. C-IMMSIM's immune simulations over three doses of the vaccine and iMODS' molecular dynamic simulations were executed to assess the reliability of the docked complexes. The stability of the vaccine construct was evaluated through the physicochemical analyses and the findings suggested that the manufactured vaccine is stable under a wide range of circumstances and can trigger immune responses against various SARS-CoV-2 variants (due to conserved epitopes). However, to strengthen the formulation of the vaccine and assess its safety and effectiveness, additional investigations and studies are required to support the computational data of this research at in-vitro and in-vivo levels.
Supplementary information: The online version contains supplementary material available at 10.1007/s40203-023-00156-2.
Keywords: Envelope protein; Immune simulation; Membrane protein; Molecular docking; Multi-epitope; Nucleocapsid; SARS-COV-2; Spike protein.
© The Author(s), under exclusive licence to Springer-Verlag GmbH Germany, part of Springer Nature 2023. Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
Conflict of interest statement
Conflict of interestThe authors declare no conflict of interest.
Figures
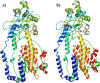


Similar articles
-
Engineering receptor-binding domain and heptad repeat domains towards the development of multi-epitopes oral vaccines against SARS-CoV-2 variants.PLoS One. 2024 Aug 15;19(8):e0306111. doi: 10.1371/journal.pone.0306111. eCollection 2024. PLoS One. 2024. PMID: 39146295 Free PMC article.
-
Designing a Multi-epitope Vaccine against the SARS-CoV-2 Variant based on an Immunoinformatics Approach.Curr Comput Aided Drug Des. 2024;20(3):274-290. doi: 10.2174/1573409919666230612125440. Curr Comput Aided Drug Des. 2024. PMID: 37309762
-
Exploring the out of sight antigens of SARS-CoV-2 to design a candidate multi-epitope vaccine by utilizing immunoinformatics approaches.Vaccine. 2020 Nov 10;38(48):7612-7628. doi: 10.1016/j.vaccine.2020.10.016. Epub 2020 Oct 9. Vaccine. 2020. PMID: 33082015 Free PMC article.
-
A reverse vaccinology and immunoinformatics approach for designing a multiepitope vaccine against SARS-CoV-2.Immunogenetics. 2021 Dec;73(6):459-477. doi: 10.1007/s00251-021-01228-3. Epub 2021 Sep 20. Immunogenetics. 2021. PMID: 34542663 Free PMC article.
-
A Bibliometric Analysis on Multi-epitope Vaccine Development Against SARS-CoV-2: Current Status, Development, and Future Directions.Mol Biotechnol. 2025 Jan 9. doi: 10.1007/s12033-024-01358-5. Online ahead of print. Mol Biotechnol. 2025. PMID: 39789401 Review.
Cited by
-
Unleashing the future of cancer immunotherapy: in silico design of a multi-epitope and mRNA vaccine duo targeting EWSR1-ATF1, EWSR1-CREB1, and PRAME to conquer clear cell sarcoma using immunoinformatics approaches.Med Oncol. 2025 Jun 28;42(8):295. doi: 10.1007/s12032-025-02863-6. Med Oncol. 2025. PMID: 40581703
-
Development of a Novel Pan-Species Multi-Epitope Vaccine (PS-MEV) Targeting Nine Staphylococcus Species to Combat Antibiotic Resistance.Probiotics Antimicrob Proteins. 2025 Apr 29. doi: 10.1007/s12602-025-10550-1. Online ahead of print. Probiotics Antimicrob Proteins. 2025. PMID: 40301233
-
Immunoinformatics design of a multi-epitope vaccine for Chlamydia trachomatis major outer membrane proteins.Sci Rep. 2024 Dec 2;14(1):29919. doi: 10.1038/s41598-024-81736-w. Sci Rep. 2024. PMID: 39623035 Free PMC article.
-
Bioinformatics and immunoinformatics approaches in the design of a multi-epitope vaccine targeting CTLA-4 for melanoma treatment.Mol Divers. 2025 Jan 28. doi: 10.1007/s11030-025-11108-7. Online ahead of print. Mol Divers. 2025. PMID: 39873886
References
-
- Alberer M, Gnad-Vogt U, Hong HS, Mehr KT, Backert L, Finak G, Gottardo R, Bica MA, Garofano A, Koch SD, Fotin-Mleczek M, Hoerr I, Clemens R, von Sonnenburg F. Safety and immunogenicity of a mRNA rabies vaccine in healthy adults: an open-label, non-randomised, prospective, first-in-human phase 1 clinical trial. Lancet. 2017;390:1511–1520. doi: 10.1016/S0140-6736(17)31665-3. - DOI - PubMed
-
- Arya H, Bhatt TK. Chapter 20 - role of bioinformatics in subunit vaccine design. In: Coumar MS, editor. Molecular docking for computer-aided drug design. UK: Academic Press; 2021. pp. 425–439.
LinkOut - more resources
Full Text Sources
Miscellaneous

