Route of dexamethasone administration influences parasite burden in Strongyloides hyperinfection model
- PMID: 37520210
- PMCID: PMC10382442
- DOI: 10.1007/s12639-023-01595-6
Route of dexamethasone administration influences parasite burden in Strongyloides hyperinfection model
Abstract
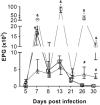
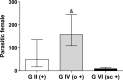
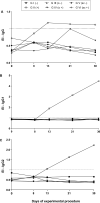
Rodents infected with Strongyloides venezuelensis are experimental models applied to strongyloidiasis research. This study evaluated oral and subcutaneous dexamethasone (DEX) treatments to establish immunosuppression in an experimental model of Strongyloides hyperinfection. Rattus norvegicus Wistar were divided: G I (-): untreated and uninfected animals, G II (+): untreated and infected, G III (o -) orally treated and uninfected, G IV (o +) orally treated and infected, G V (sc -) subcutaneously treated and uninfected, G VI (sc +) subcutaneously treated and infected. For oral administration, DEX was diluted in sterile water (5 µg/ml) and made available to the animals on intervals in experimental days - 5-0, 8-13 and 21-26. For subcutaneous administration, animals received daily injections of DEX disodium phosphate (2 mg/kg). Infection was established by the subcutaneous inoculation of 3000 S. venezuelensis filarioid larvae. Groups were evaluated by egg per gram of feces and parasite females counts and IgG, IgG1 and IgG2a detection. GIV (o +) had egg peaks count on days 13 and 26 and maintained egg elimination until the last experimental day. Parasitic females recovery at day 30 was significantly higher in G IV (o +) when compared to G VI (sc +). Levels of IgG, IgG1 and IgG2a of all groups, except the positive control GII (+), were below the detection threshold. Pharmacological immunosuppression induced by oral administration of DEX produced high parasitic burden, and is a noninvasive method, useful to establish immunosuppression in strongyloidiasis hyperinfection model in rats.
Keywords: Dexamethasone; Experimental infection; Hyperinfection; Immunosuppression; Strongyloides venezuelensis.
© Indian Society for Parasitology 2023. Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
Conflict of interest statement
Conflict of interestThe authors declare no competing interests.
Figures
Similar articles
-
Parasitological and immunological aspects of oral and subcutaneous prednisolone treatment in rats experimentally infected with Strongyloides venezuelensis.Acta Trop. 2020 Apr;204:105349. doi: 10.1016/j.actatropica.2020.105349. Epub 2020 Jan 17. Acta Trop. 2020. PMID: 31958413
-
Treatment with nitric oxide donors diminishes hyperinfection by Strongyloides venezuelensis in mice treated with dexamethasone.Acta Trop. 2015 Dec;152:90-95. doi: 10.1016/j.actatropica.2015.08.019. Epub 2015 Sep 2. Acta Trop. 2015. PMID: 26342794
-
Strongyloides venezuelensis infection augments arterial blood pressure in male wistar rats.Acta Trop. 2019 Feb;190:350-355. doi: 10.1016/j.actatropica.2018.12.007. Epub 2018 Dec 6. Acta Trop. 2019. PMID: 30529092
-
Strongyloides infection in rodents: immune response and immune regulation.Parasitology. 2017 Mar;144(3):295-315. doi: 10.1017/S0031182016000111. Epub 2016 Feb 24. Parasitology. 2017. PMID: 26905057 Review.
-
Metaperiodate deglycosylation of Strongyloides venezuelensis larvae: Immunochemical characterization and antigen production for human strongyloidiasis diagnosis.Acta Trop. 2018 Jun;182:27-33. doi: 10.1016/j.actatropica.2018.02.001. Epub 2018 Feb 15. Acta Trop. 2018. PMID: 29454735 Review.
References
-
- Carvalho EFG, Neto de Sousa JE, Gonçalves AL, da Cunha-Junior JP, Costa-Cruz JM. Immunoblotting using Strongyloides venezuelensis larvae, parthenogenetic females or eggs extracts for the diagnosis of experimentally infected immunosuppressed rats. Exp Parasitol. 2015;157:117–123. doi: 10.1016/j.exppara.2015.07.009. - DOI - PubMed
-
- Chaves LA, Gonçalves AL, Paula FM, Silva NM, Silva CV, Costa-Cruz JM, Freitas MA. Comparison of parasitological, immunological and molecular methods for evaluation of fecal samples of immunosuppressed rats experimentally infected with Strongyloides venezuelensis. Parasitol. 2015;142(14):1715–1721. doi: 10.1017/S0031182015001298. - DOI - PubMed
LinkOut - more resources
Full Text Sources
