Early protective effect of a ("pan") coronavirus vaccine (PanCoVac) in Roborovski dwarf hamsters after single-low dose intranasal administration
- PMID: 37520530
- PMCID: PMC10372429
- DOI: 10.3389/fimmu.2023.1166765
Early protective effect of a ("pan") coronavirus vaccine (PanCoVac) in Roborovski dwarf hamsters after single-low dose intranasal administration
Abstract
Introduction: The coronavirus disease 2019 (COVID-19) pandemic caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has highlighted the danger posed by human coronaviruses. Rapid emergence of immunoevasive variants and waning antiviral immunity decrease the effect of the currently available vaccines, which aim at induction of neutralizing antibodies. In contrast, T cells are marginally affected by antigen evolution although they represent the major mediators of virus control and vaccine protection against virus-induced disease.
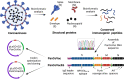
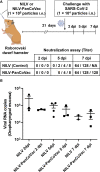
Materials and methods: We generated a multi-epitope vaccine (PanCoVac) that encodes the conserved T cell epitopes from all structural proteins of coronaviruses. PanCoVac contains elements that facilitate efficient processing and presentation of PanCoVac-encoded T cell epitopes and can be uploaded to any available vaccine platform. For proof of principle, we cloned PanCoVac into a non-integrating lentivirus vector (NILV-PanCoVac). We chose Roborovski dwarf hamsters for a first step in evaluating PanCoVac in vivo. Unlike mice, they are naturally susceptible to SARS-CoV-2 infection. Moreover, Roborovski dwarf hamsters develop COVID-19-like disease after infection with SARS-CoV-2 enabling us to look at pathology and clinical symptoms.
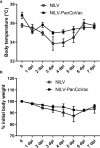
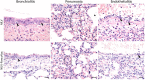
Results: Using HLA-A*0201-restricted reporter T cells and U251 cells expressing a tagged version of PanCoVac, we confirmed in vitro that PanCoVac is processed and presented by HLA-A*0201. As mucosal immunity in the respiratory tract is crucial for protection against respiratory viruses such as SARS-CoV-2, we tested the protective effect of single-low dose of NILV-PanCoVac administered via the intranasal (i.n.) route in the Roborovski dwarf hamster model of COVID-19. After infection with ancestral SARS-CoV-2, animals immunized with a single-low dose of NILV-PanCoVac i.n. did not show symptoms and had significantly decreased viral loads in the lung tissue. This protective effect was observed in the early phase (2 days post infection) after challenge and was not dependent on neutralizing antibodies.
Conclusion: PanCoVac, a multi-epitope vaccine covering conserved T cell epitopes from all structural proteins of coronaviruses, might protect from severe disease caused by SARS-CoV-2 variants and future pathogenic coronaviruses. The use of (HLA-) humanized animal models will allow for further efficacy studies of PanCoVac-based vaccines in vivo.
Keywords: T cell epitopes; T-cell-directed vaccine; coronaviruses; dwarf hamster COVID-19 model; multi-epitope vaccine; pan-coronavirus vaccine; universal COVID-19 vaccine.
Copyright © 2023 Abdelaziz, Raftery, Weihs, Bielawski, Edel, Köppke, Vladimirova, Adler, Firsching, Voß, Gruber, Hummel, Fernandez Munoz, Müller-Marquardt, Willimsky, Elleboudy, Trimpert and Schönrich.
Conflict of interest statement
MA, MR and GS hold a patent EP4176898 related to the PanCoVac vaccine described herein. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
Publication types
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

