Randomized clinical trial examining financial incentives for smoking cessation among mothers of young children and possible impacts on child secondhand smoke exposure
- PMID: 37527730
- PMCID: PMC10825067
- DOI: 10.1016/j.ypmed.2023.107651
Randomized clinical trial examining financial incentives for smoking cessation among mothers of young children and possible impacts on child secondhand smoke exposure
Abstract
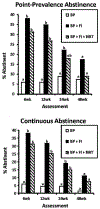
This randomized clinical trial examined whether financial-incentives increase smoking cessation among mothers of young children and potential impacts on child secondhand-smoke exposure (SHSe). 198 women-child dyads were enrolled and assigned to one of three treatment conditions: best practices (BP, N = 68), best practices plus financial incentives (BP + FI, N = 63), or best practices, financial incentives, and nicotine replacement therapy (BP + FI + NRT, N = 67). The trial was completed in Vermont, USA between June 2015 and October 2020. BP entailed staff referral to the state tobacco quitline; financial incentives entailed mothers earning vouchers exchangeable for retail items for 12 weeks contingent on biochemically-verified smoking abstinence; NRT involved mothers receiving 10 weeks of free transdermal nicotine and nicotine lozenges/gum. Baseline, 6-, 12-, 24-, and 48-week assessments were conducted. Primary outcomes were maternal 7-day point-prevalence abstinence and child SHSe through the 24-week assessment with the 48-week assessment exploratory. Results were analyzed using mixed model repeated measures for categorical data. Odds of maternal abstinence were greater among mothers in BP + FI and BP + FI + NRT compared to BP at the 6- and 12-week assessments (ORs ≥ 7.30; 95% CIs: 2.35-22.71); only abstinence in BP + FI remained greater than BP at the 24-week assessment (OR = 2.95; 95% CIs: 1.06-8.25). Abstinence did not differ significantly between treatment conditions at the 48-week assesssment. There was a significant effect of treatment condition (F[2109] = 3.64, P = .029) on SHSe with levels in BP and BP + FI significantly below BP + FI + NRT (ts[109] ≥ -2.30, Ps ≤ 0.023). Financial incentives for smoking abstinence are efficacious for increasing maternal cessation but that alone was insufficient for reducing child SHSe. ClinicalTrials.gov:NCT05740098.
Keywords: Best practices; Financial incentives; Mothers; Nicotine replacement therapy; Secondhand smoke exposure; Smoking cessation; Women.
Copyright © 2023. Published by Elsevier Inc.
Conflict of interest statement
Declaration of Competing Interest The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures

References
-
- Armitage P, 1983. Exclusions, losses to follow-up, and withdrawals in clinical trials. In: Shapiro SH, Lewis TA (Eds.), Clinical trials: Issues and approaches. Marcel Dekker, Inc, New York, pp. 99–113.
-
- Brown RA, Burgess ES, Sales SD, Whiteley JA, Evans DM, & Miller IW, 1998. Reliability and validity of a smoking timeline follow-back interview. Psychol Addict Behav, 12, 101–12. 10.1037/0893-164X.12.2.101. - DOI
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

