Rational application of EGFR-TKI adjuvant therapy in patients with completely resected stage IB-IIIA EGFR-mutant NSCLC: a systematic review and meta-analysis of 11 randomized controlled trials
- PMID: 37528390
- PMCID: PMC10391763
- DOI: 10.1186/s12885-023-11194-6
Rational application of EGFR-TKI adjuvant therapy in patients with completely resected stage IB-IIIA EGFR-mutant NSCLC: a systematic review and meta-analysis of 11 randomized controlled trials
Abstract
Purpose: To determine the role and rational application of epidermal growth factor receptor tyrosine kinase inhibitor (EGFR-TKI) adjuvant therapy in patients with completely resected stage IB-IIIA EGFR-mutant non-small-cell lung cancer (NSCLC).
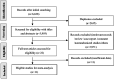
Method: Randomized controlled trials (RCTs) that compared the survival outcomes between adjuvant EGFR-TKIs and adjuvant chemotherapy or a placebo, or between different EGFR-TKI treatment durations for resected NSCLC, were eligible for inclusion. Disease-free survival (DFS) and overall survival (OS) with hazard ratios (HRs) and 95% confidence intervals (CIs) were calculated as effective measures using random-effect or fixed-effect models. Subgroup analysis was also performed.
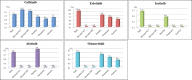
Results: Eleven RCTs involving 2102 EGFR-mutant NSCLC patients with or without EGFR-TKI adjuvant therapy were included. For all stage IB-IIIA NSCLC patients, EGFR-TKIs adjuvant therapy could not only significantly improve DFS (HR 0.43, 95% CI 0.30-0.63, P < 0.001) and 2- and 3-year DFS rates, but also improve OS (HR 0.72, 95% CI, 0.54-0.96, P = 0.024), compared with chemotherapy or the placebo. Further subgroup analyses indicated prolonged OS from first-generation EGFR-TKI adjuvant therapy in stage III patients, compared with chemotherapy or the placebo (HR for OS, 0.34; 95% CI, 0.18-0.63; P = 0.001). Of note, osimertinib adjuvant therapy led to the OS benefit expanding from stage III to stage II-III patients, with significantly improved DFS and a lower risk of brain recurrence, compared with the placebo. A 2-year treatment duration with EGFR-TKI adjuvant therapy showed a significantly lower recurrence risk than a ≤ 1-year duration.
Conclusion: The DFS advantage from first-generation EGFR-TKI adjuvant therapy can translate into an OS benefit in stage III NSCLC patients. Osimertinib might be more suitable for adjuvant therapy than first-generation EGFR-TKIs, because of the lower recurrence rate and the potential OS benefit even in early-stage patients. The optimal treatment duration for EGFR-TKIs at different stages of disease needs to be validated.
Keywords: Adjuvant therapy; Epidermal growth factor receptor tyrosine kinase inhibitor; Meta-analysis; Mutation; Non-small-cell lung cancer.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures




References
-
- Kris MG, Gaspar LE, Chaft JE, Kennedy EB, Azzoli CG, Ellis PM, Lin SH, Pass HI, Seth R, Shepherd FA, Spigel DR, Strawn JR, Ung YC, Weyant M. Adjuvant systemic therapy and adjuvant Radiation Therapy for Stage I to IIIA completely resected non-small-cell lung cancers: American Society of Clinical Oncology/Cancer Care Ontario Clinical Practice Guideline Update. J Clin Oncol. 2017;35:2960–74. doi: 10.1200/JCO.2017.72.4401. - DOI - PubMed
-
- Pignon JP, Tribodet H, Scagliotti GV, Douillard JY, Shepherd FA, Stephens RJ, Dunant A, Torri V, Rosell R, Seymour L, Spiro SG, Rolland E, Fossati R, Aubert D, Ding K, Waller D, Le Chevalier T, Group LC. Lung adjuvant cisplatin evaluation: a pooled analysis by the LACE Collaborative Group. J Clin Oncol. 2008;26:3552–9. doi: 10.1200/JCO.2007.13.9030. - DOI - PubMed
-
- Ciardiello F, Tortora G. A novel approach in the treatment of cancer: targeting the epidermal growth factor receptor. Clin Cancer Res. 2001;7:2958–70. - PubMed
-
- NCCN clinical practice guidelines. In Oncology:Non-Small Cell Lung Cancer(2022.V5) https://www.nccn.org/professionals/physician_gls/default.aspx [.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

