Mirvetuximab Soravtansine in Platinum-Resistant Ovarian Cancer
- PMID: 37528890
- PMCID: PMC10387675
- DOI: 10.1177/11795549231187264
Mirvetuximab Soravtansine in Platinum-Resistant Ovarian Cancer
Abstract
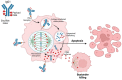
Ovarian cancer is the second leading cause of death from gynecologic malignancies worldwide. Management of platinum-resistant disease is challenging and clinical outcomes with standard chemotherapy are poor. Over the past decades, significant efforts have been made to understand drug resistance and develop strategies to overcome treatment failure. Antibody drug conjugates (ADCs) are a rapidly growing class of oncologic therapeutics, which combine the ability to target tumor-specific antigens with the cytotoxic effects of chemotherapy. Mirvetuximab soravtansine is an ADC comprising an IgG1 monoclonal antibody against the folate receptor alpha (FRα) conjugated to the cytotoxic maytansinoid effector molecule DM4 that has shown promising clinical activity in patients with FR-α-positive ovarian cancer. This review summarizes current evidence of mirvetuximab soravtansine in platinum-resistant ovarian cancer, focusing on clinical activity, toxicity, and future directions.
Keywords: Antibody drug conjugate; folate receptor; mirvetuximab; ovarian cancer; platinum resistance.
© The Author(s) 2023.
Conflict of interest statement
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: EG reports no conflict of interest. AV reports no conflict of interest; AO reports no conflict of interest; PI and Steering Committees with AstraZeneca, GSK, and Clovis. Advisory Boards AstraZeneca and Morphosys.
Figures
References
-
- Sung H, Ferlay J, Siegel RL, et al.. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71:209-249. - PubMed
-
- Prat J. Ovarian carcinomas: five distinct diseases with different origins, genetic alterations, and clinicopathological features. Virchows Arch. 2012;460:237-249. - PubMed
-
- Stuart GC, Kitchener H, Bacon M, et al.. 2010 Gynecologic Cancer InterGroup (GCIG) consensus statement on clinical trials in ovarian cancer: report from the Fourth Ovarian Cancer Consensus Conference. Int J Gynecol Cancer. 2011;21:750-755. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous