C(sp2)-H Hydroxylation via Catalytic 1,4-Ni Migration with N2O
- PMID: 37531410
- PMCID: PMC10586377
- DOI: 10.1021/jacs.3c07018
C(sp2)-H Hydroxylation via Catalytic 1,4-Ni Migration with N2O
Abstract
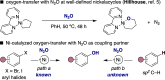
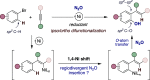
Herein, we report a Ni-catalyzed C(sp2)-H hydroxylation of aryl bromides with N2O as an oxygen-atom donor. The reaction is enabled by a 1,4-Ni translocation that results in ipso/ortho difunctionalized products. Regioselectivity and stereocontrol are dictated by a judicious choice of the ligand backbone, thus giving access to either carbonyl or phenol derivatives and offering an opportunity to repurpose hazardous substances en route to valuable oxygen-containing building blocks.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
Similar articles
-
Nickel-Catalyzed Ipso/Ortho Difunctionalization of Aryl Bromides with Alkynes and Alkyl Bromides via a Vinyl-to-Aryl 1,4-Hydride Shift.J Am Chem Soc. 2021 Dec 8;143(48):20064-20070. doi: 10.1021/jacs.1c10368. Epub 2021 Nov 18. J Am Chem Soc. 2021. PMID: 34793680
-
Native Amides as Enabling Vehicles for Forging sp3-sp3 Architectures via Interrupted Deaminative Ni-Catalyzed Chain-Walking.J Am Chem Soc. 2023 Feb 22;145(7):3869-3874. doi: 10.1021/jacs.2c12915. Epub 2023 Feb 8. J Am Chem Soc. 2023. PMID: 36753384 Free PMC article.
-
Synthetic and Mechanistic Implications of Chlorine Photoelimination in Nickel/Photoredox C(sp3)-H Cross-Coupling.Acc Chem Res. 2021 Feb 16;54(4):988-1000. doi: 10.1021/acs.accounts.0c00694. Epub 2021 Jan 29. Acc Chem Res. 2021. PMID: 33511841 Free PMC article.
-
Transition Metal-Catalyzed Regioselective Direct C-H Amidation: Interplay between Inner- and Outer-Sphere Pathways for Nitrene Cross-Coupling Reactions.Acc Chem Res. 2022 Aug 2;55(15):2123-2137. doi: 10.1021/acs.accounts.2c00283. Epub 2022 Jul 19. Acc Chem Res. 2022. PMID: 35853135
-
Ni-Catalyzed Oxygen Transfer from N2O onto sp3-Hybridized Carbons.J Am Chem Soc. 2022 Oct 12;144(40):18223-18228. doi: 10.1021/jacs.2c06227. Epub 2022 Sep 26. J Am Chem Soc. 2022. PMID: 36162124 Free PMC article.
Cited by
-
Hydroxylation of Aryl Sulfonium Salts for Phenol Synthesis under Mild Reaction Conditions.Molecules. 2024 Feb 13;29(4):831. doi: 10.3390/molecules29040831. Molecules. 2024. PMID: 38398583 Free PMC article.
-
Nitrous oxide as diazo transfer reagent.Chem Sci. 2024 Aug 14;15(34):13605-17. doi: 10.1039/d4sc04530k. Online ahead of print. Chem Sci. 2024. PMID: 39156938 Free PMC article. Review.
-
Cu-Promoted ipso-Hydroxylation of sp2 Bonds with Concomitant Aromatic 1,2-Rearrangement Involving a Cu-oxyl-hydroxo Species.Inorg Chem. 2024 Oct 28;63(43):20675-20688. doi: 10.1021/acs.inorgchem.4c03304. Epub 2024 Oct 18. Inorg Chem. 2024. PMID: 39422540 Free PMC article.
-
Rapid Oxygen Atom Transfer at a Catalysis-Relevant Ni(I)-Alkyl Complex with N2O.J Am Chem Soc. 2025 Jun 11;147(23):19438-19443. doi: 10.1021/jacs.5c03351. Epub 2025 May 29. J Am Chem Soc. 2025. PMID: 40442965 Free PMC article.
References
-
- Prather M. J. Time Scales in Atmospheric Chemistry: Coupled Perturbations to N2O, NOy, and O3. Science 1998, 279, 1339–1341. 10.1126/science.279.5355.1339. - DOI - PubMed
- Ravishankara A. R.; Daniel J. S.; Portmann R. W. Nitrous Oxide (N2O): The Dominant Ozone-Depleting Substance Emitted in the 21st Century. Science 2009, 326, 123–125. 10.1126/science.1176985. - DOI - PubMed
- Prather M. J.; Hsu J. Coupling of Nitrous Oxide and Methane by Global Atmospheric Chemistry. Science 2010, 330, 952–954. 10.1126/science.1196285. - DOI - PubMed
- Tian H.; Xu R.; Canadell J. G.; Thompson R. L.; Winiwarter W.; Suntharalingam P.; Davidson E. A.; Ciais P.; Jackson R. B.; Janssens-Maenhout G.; Prather M. J.; Regnier P.; Pan N.; Pan S.; Peters G. P.; Shi H.; Tubiello F. N.; Zaehle S.; Zhou F.; Arneth A.; Battaglia G.; Berthet S.; Bopp L.; Bouwman A. F.; Buitenhuis E. T.; Chang J.; Chipperfield M. P.; Dangal S. R. S.; Dlugokencky E.; Elkins J. W.; Eyre B. D.; Fu B.; Hall B.; Ito A.; Joos F.; Krummel P. B.; Landolfi A.; Laruelle G. G.; Lauerwald R.; Li W.; Lienert S.; Maavara T.; MacLeod M.; Millet D. B.; Olin S.; Patra P. K.; Prinn R. G.; Raymond P. A.; Ruiz D. J.; van der Werf G. R.; Vuichard N.; Wang J.; Weiss R. F.; Wells K. C.; Wilson C.; Yang J.; Yao Y. A Comprehensive Quantification of Global Nitrous Oxide Sources and Sinks. Nature 2020, 586, 248–256. 10.1038/s41586-020-2780-0. - DOI - PubMed
-
- For a selection of techniques that repurpose N2O en route to valuable added-value chemicals, seeYamada T.; Hashimoto K.; Kitaichi Y.; Suzuki K.; Ikeno T. Nitrous Oxide Oxidation of Olefins Catalyzed by Ruthenium Porphyrin Complexes. Chem. Lett. 2001, 30, 268–269. 10.1246/cl.2001.268. - DOI
- Tskhovrebov A. G.; Solari E.; Scopelliti R.; Severin K. Organometallics 2014, 33, 2405–2408. 10.1021/om500333y. - DOI
- Kiefer G.; Riedel T.; Dyson P. J.; Scopelliti R.; Severin K. Synthesis of Triazenes with Nitrous Oxide. Angew. Chem., Int. Ed. 2015, 54, 302–305. 10.1002/anie.201408597. - DOI - PubMed
- Banert K.; Singh N.; Fiedler B.; Friedrich J.; Korb M.; Lang H. 4,5-Dihydro-1,2,3-oxadiazole: A Very Elusive Key Intermediate in Various Important Chemical Transformations. Chem.—Eur. J. 2015, 21, 15092–15099. 10.1002/chem.201502326. - DOI - PubMed
- Gianetti T. L.; Annen S. P.; Santiso-Quinones G.; Reiher M.; Driess M.; Grützmacher H. Nitrous Oxide as a Hydrogen Acceptor for the Dehydrogenative Coupling of Alcohols. Angew. Chem., Int. Ed. 2016, 55, 1854–1858. 10.1002/anie.201509288. - DOI - PubMed
- Gianetti T. L.; Rodríguez-Lugo R. E.; Harmer J. R.; Trincado M.; Vogt M.; Santiso-Quinones G.; Grützmacher H. Zero-Valent Amino-Olefin Cobalt Complexes as Catalysts for Oxygen Atom Transfer Reactions from Nitrous Oxide. Angew. Chem., Int. Ed. 2016, 55, 15323–15328. 10.1002/anie.201609173. - DOI - PubMed
- Zeng R.; Feller M.; Ben-David Y.; Milstein D. Hydrogenation and Hydrosilylation of Nitrous Oxide Homogeneously Catalyzed by a Metal Complex. J. Am. Chem. Soc. 2017, 139, 5720–5723. 10.1021/jacs.7b02124. - DOI - PMC - PubMed
- Zeng R.; Feller M.; Diskin-Posner Y.; Shimon L. J. W.; Ben-David Y.; Milstein D. CO Oxidation by N2O Homogeneously Catalyzed by Ruthenium Hydride Pincer Complexes Indicating a New Mechanism. J. Am. Chem. Soc. 2018, 140, 7061–7064. 10.1021/jacs.8b03927. - DOI - PMC - PubMed
- Eymann L. Y. M.; Varava P.; Shved A. M.; Curchod B. F. E.; Liu Y.; Planes M. O.; Sienkiewicz A.; Scopelliti R.; Tirani F. F.; Severin K. Synthesis of Organic Super-Electron-Donors by Reaction of Nitrous Oxide with N-Heterocyclic Olefins. J. Am. Chem. Soc. 2019, 141, 17112–17116. 10.1021/jacs.9b10660. - DOI - PubMed
-
- Tolman W. B. Binding and Activation of N2O at Transition-Metal Centers: Recent Mechanistic Insights. Angew. Chem., Int. Ed. 2010, 49, 1018–1024. 10.1002/anie.200905364. - DOI - PMC - PubMed
- Figg T. M.; Cundari T. R. Mechanistic Study of Oxy Insertion into Nickel-Carbon Bonds with Nitrous Oxide. Organometallics 2012, 31, 4998–5004. 10.1021/om300270x. - DOI
- Yao L.; Li Y.; Huang L.; Guo K.; Ren G.; Wu Z.; Lei Q.; Fang W.; Xie H. A DFT Study on the Mechanisms of Hydrogenation and Hydrosilylation of Nitrous Oxide Catalyzed by a Ruthenium PNP Pincer Complex. Comput. Theor. Chem. 2018, 1128, 48–55. 10.1016/j.comptc.2018.02.010. - DOI
- Lehnert N.; Dong H. T.; Harland J. B.; Hunt A. P.; White C. J. Reversing Nitrogen Fixation. Nat. Rev. Chem. 2018, 2, 278–289. 10.1038/s41570-018-0041-7. - DOI
-
- Matsunaga P. T.; Hillhouse G. L.; Rheingold A. L. Oxygen-Atom Transfer from Nitrous Oxide to a Nickel Metallacycle. Synthesis, Structure, and Reactions of [Cyclic] (2,2’-Bipyridine) Ni(OCH2CH2CH2CH2). J. Am. Chem. Soc. 1993, 115, 2075–2077. 10.1021/ja00058a085. - DOI
- Koo K.; Hillhouse G. L.; Rheingold A. L. Oxygen-Atom Transfer from Nitrous Oxide to an Organonickel(II) Phosphine Complex. Syntheses and Reactions of New Nickel(II) Aryloxides and the Crystal Structure of [Cyclic] (Me2PCH2CH2PMe2) Ni(O-o-C6H4CMe2CH2). Organometallics 1995, 14, 456–460. 10.1021/om00001a062. - DOI
- Matsunaga P. T.; Mavropoulos J. C.; Hillhouse G. L. Oxygen-Atom Transfer from Nitrous Oxide (N N O) to Nickel Alkyls. Syntheses and Reactions of Nickel(II) Alkoxides. Polyhedron 1995, 14, 175–185. 10.1016/0277-5387(94)00330-H. - DOI
LinkOut - more resources
Full Text Sources
Research Materials