Disruptive 3D in vitro models for respiratory disease investigation: A state-of-the-art approach focused on SARS-CoV-2 infection
- PMID: 37534107
- PMCID: PMC10391659
- DOI: 10.1016/j.bbiosy.2023.100082
Disruptive 3D in vitro models for respiratory disease investigation: A state-of-the-art approach focused on SARS-CoV-2 infection
Abstract
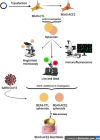
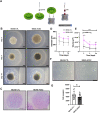
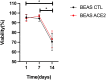
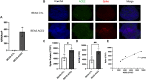
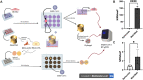
COVID-19, along with most respiratory diseases in the medical field, demonstrates significant ability to take its toll on global population. There is a particular difficulty in studying these conditions, which stems especially from the short supply of in vitro models for detailed investigation, the specific therapeutic knowledge required for disease scrutinization and the occasional need of BSL-3 [Biosafety Level 3] laboratories for research. Based on this, the process of drug development is hampered to a great extent. In the scenario of COVID-19, this difficulty is even more substantial on account of the current undefinition regarding the exact role of the ACE2 [Angiotensin-converting enzyme 2] receptor upon SARS-CoV-2 kinetics in human cells and the great level of demand in the investigation process of ACE2, which usually requires the laborious and ethically complicated usage of transgenic animal models overexpressing the receptor. Moreover, the rapid progression of the aforementioned diseases, especially COVID-19, poses a crucial necessity for adequate therapeutic solutions emergence. In this context, the work herein presented introduces a groundbreaking set of 3D models, namely spheroids and MatriWell cell culture inserts, whose remarkable ability to mimic the in vivo environment makes them highly suitable for respiratory diseases investigation, particularly SARS-CoV-2 infection. Using MatriWells, we developed an innovative platform for COVID-19 research: a pulmonary air-liquid interface [ALI] associated with endothelial (HUVEC) cells. Infection studies revealed that pulmonary (BEAS-2B) cells in the ALI reached peak viral load at 24h and endothelial cells, at 48h, demonstrating lung viral replication and subsequent hematogenous dissemination, which provides us with a unique and realistic framework for studying COVID-19. Simultaneously, the spheroids were used to address the understudied ACE2 receptor, aiming at a pronounced process of COVID-19 investigation. ACE2 expression not only increased spheroid diameter by 20% (p<0.001) and volume by 60% (p≤0.0001) but also led to a remarkable 640-fold increase in intracellular viral load (p≤0.01). The previously mentioned finding supports ACE2 as a potential target for COVID-19 treatment. Lastly, we observed a higher viral load in the MatriWells compared to spheroids (150-fold, p<0.0001), suggesting the MatriWells as a more appropriate approach for COVID-19 investigation. By establishing an advanced method for respiratory tract conditions research, this work paves the way toward an efficacious process of drug development, contributing to a change in the course of respiratory diseases such as COVID-19.
Keywords: 3D culture; ACE2; COVID-19; MatriWell; Respiratory diseases; SARS-CoV-2.
© 2023 The Author(s).
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper. Gabriel Romero Liguori and Tácia Tavares Aquinas Liguori are, respectively, the Chief Executive Officer and the Chief Scientific Officer of TissueLabs Sagl (Manno, Switzerland), the company which developed and produced MatriWell cell culture inserts. The remaining authors have no conflict of interest to declare.
Figures

Similar articles
-
SARS-CoV-2 infection and replication kinetics in different human cell types: The role of autophagy, cellular metabolism and ACE2 expression.Life Sci. 2022 Nov 1;308:120930. doi: 10.1016/j.lfs.2022.120930. Epub 2022 Sep 6. Life Sci. 2022. PMID: 36075471 Free PMC article.
-
Human Air-Liquid-Interface Organotypic Airway Cultures Express Significantly More ACE2 Receptor Protein and Are More Susceptible to HCoV-NL63 Infection than Monolayer Cultures of Primary Respiratory Epithelial Cells.Microbiol Spectr. 2022 Aug 31;10(4):e0163922. doi: 10.1128/spectrum.01639-22. Epub 2022 Jul 12. Microbiol Spectr. 2022. PMID: 35863002 Free PMC article.
-
Testing the efficacy and safety of BIO101, for the prevention of respiratory deterioration, in patients with COVID-19 pneumonia (COVA study): a structured summary of a study protocol for a randomised controlled trial.Trials. 2021 Jan 11;22(1):42. doi: 10.1186/s13063-020-04998-5. Trials. 2021. PMID: 33430924 Free PMC article.
-
ACE2: from protection of liver disease to propagation of COVID-19.Clin Sci (Lond). 2020 Dec 11;134(23):3137-3158. doi: 10.1042/CS20201268. Clin Sci (Lond). 2020. PMID: 33284956 Review.
-
Role of angiotensin-converting enzyme 2 and pericytes in cardiac complications of COVID-19 infection.Am J Physiol Heart Circ Physiol. 2020 Nov 1;319(5):H1059-H1068. doi: 10.1152/ajpheart.00681.2020. Epub 2020 Oct 9. Am J Physiol Heart Circ Physiol. 2020. PMID: 33036546 Free PMC article. Review.
Cited by
-
Antiviral, anti-inflammatory and antioxidant effects of curcumin and curcuminoids in SH-SY5Y cells infected by SARS-CoV-2.Sci Rep. 2024 May 10;14(1):10696. doi: 10.1038/s41598-024-61662-7. Sci Rep. 2024. PMID: 38730068 Free PMC article.
References
-
- Coronavirus Resource Center, Johns Hopkins University. https://coronavirus.jhu.edu/map.html, 2023 (accessed 01 February 2023).
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
