Real-world effectiveness, satisfaction, and optimization of ubrogepant for the acute treatment of migraine in combination with onabotulinumtoxinA: results from the COURAGE Study
- PMID: 37537578
- PMCID: PMC10399003
- DOI: 10.1186/s10194-023-01622-0
Real-world effectiveness, satisfaction, and optimization of ubrogepant for the acute treatment of migraine in combination with onabotulinumtoxinA: results from the COURAGE Study
Abstract
Background: Individuals using onabotulinumtoxinA as a preventive migraine treatment often use acute treatments for breakthrough attacks. Data on real-world effectiveness of the small-molecule calcitonin gene-related peptide (CGRP) receptor antagonist ubrogepant in combination with onabotulinumtoxinA are limited.
Methods: COURAGE, a prospective, multiple attack, observational study, evaluated the real-world effectiveness of ubrogepant (50 or 100 mg) for acute treatment of migraine in people receiving onabotulinumtoxinA, an anti-CGRP monoclonal antibody (mAb), or both. This analysis focused only on onabotulinumtoxinA users. The Migraine Buddy app was used to identify eligible participants and track response to treated attacks. For each ubrogepant-treated attack, meaningful pain relief (MPR) and return to normal function (RNF) at 2 and 4 h post-dose over 30 days was assessed. MPR was defined as a level of relief that is meaningful to the participant, usually occurring before the pain is all gone. After 30 days, satisfaction was reported on a 7-point scale and overall acute treatment optimization was evaluated using the migraine Treatment Optimization Questionnaire-4 (mTOQ-4).
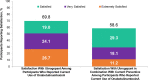
Results: This analysis included 122 participants who received ubrogepant and onabotulinumtoxinA and reported on 599 ubrogepant-treated attacks. Following the first ubrogepant-treated attack, MPR was achieved in 53.3% of participants 2 h post-dose and in 76.2% of participants 4 h post-dose. RNF was achieved in 25.4% of participants 2 h post-dose and in 45.9% of participants 4 h post-dose. MPR and RNF results were similar across up to 10 ubrogepant-treated attacks. After 30 days, satisfaction with ubrogepant in combination with onabotulinumtoxinA was reported by 69.8% of participants and acute treatment optimization (defined as mTOQ-4 score ≥ 4) was achieved in 77.6%.
Conclusions: In this prospective real-world effectiveness study, ubrogepant treatment in onabotulinumtoxinA users with self-identified migraine was associated with high rates of MPR and RNF at 2 and 4 h as well as satisfaction and acute treatment optimization. Although the lack of a contemporaneous control group limits causal inference, these findings demonstrate the feasibility of using a novel, app-based design to evaluate the real-world effectiveness and satisfaction of treatments.
Keywords: Chronic migraine; OnabotulinumtoxinA; Ubrogepant.
© 2023. Springer-Verlag Italia S.r.l., part of Springer Nature.
Conflict of interest statement
RBL has received research support from the National Institutes of Health, the FDA, and the National Headache Foundation. He serves as consultant, advisory board member, or has received honoraria or research support from AbbVie/Allergan, Amgen, Biohaven, Dr. Reddy’s Laboratories (Promius), electroCore, Eli Lilly, GlaxoSmithKline, Lundbeck, Merck, Novartis, Teva, Vector, and Vedanta Research. He receives royalties from
EE, NDA, and DS were employees of OPEN Health Group at the time of study conduct.
LD serves as a consultant, advisory board member, or has received honoraria from AbbVie, Amgen, Eli Lilly, Novartis, and Teva.
AMA, JC-DL, and KS are employees of AbbVie and may hold AbbVie stock.
SH serves as a consultant, advisory board member, or has received honoraria from AbbVie, Amgen, Biohaven, Currax, Eli Lilly, Impel, Lundbeck, Novartis, Teva, Theranica, and Upsher-Smith.
Figures



References
-
- Steiner TJ, Jensen R, Katsarava Z, Linde M, MacGregor EA, Osipova V, et al. Aids to management of headache disorders in primary care (2nd edition): on behalf of the European Headache Federation and Lifting the Burden: the Global Campaign against Headache. J Headache Pain. 2019;20:57. doi: 10.1186/s10194-018-0899-2. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

